A Brief Overview of Tyndall Effect and Scattering of Light
The Tyndall Effect and Scattering of Light are optical phenomena involving light interacting with particles in a medium. The Tyndall Effect occurs when light is scattered all around by small particles or colloidal suspensions, making its path visible. It’s sensitive to particle size and concentration levels and finds applications across fields like chemistry and medicine, helping identify specific characteristics in particles.
Scattering of Light, on the other hand, is an umbrella term encompassing various scattering processes. Rayleigh scattering explains why shorter wavelengths scatter more. Mie scattering in larger particles and clouds often results in halos and sunset hues; understanding this phenomenon is crucial to atmospheric science, astronomy, and particle characterization techniques.
While the Tyndall Effect is one specific instance of light scattering in colloidal suspensions, scattering can refer to various mechanisms with various applications in both scientific and everyday life. Both phenomena demonstrate how light interacts with matter to shape our understanding of our environment and how we view ourselves within it.
What is Tyndall Effect?
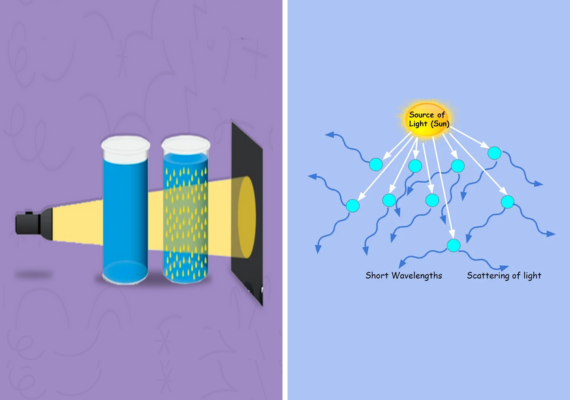
The Tyndall Effect, named for 19th-century British scientist John Tyndall, is an optical and colloid chemistry phenomenon in which light scatters or disperses in various directions when passing through a medium with small particles or colloidal suspensions, such as colloids. This scattering makes the path of light visible as it is scattered in various directions creating a cone of illumination visible against any background light source.

The Tyndall Effect becomes most evident when particles in the medium are roughly similar in size to its wavelength and create a visible cone of illumination from within a medium that contains particles roughly equivalent to its wavelength.
The Tyndall Effect involves light scattering when photons of light interact with particles in a medium, causing photons of light to change direction and scatter due to differences in refractive index between these particles and their surroundings.
As such, shorter wavelengths (such as blue and violet light rays ) tend to be scattered more than longer ones such as red and yellow wavelengths resulting in a visible cone of light appearing bluish in hue.
The Tyndall Effect can be utilized in numerous fields. Chemists use it to detect colloids in solutions and to quantify their concentration; medical professionals use the Tyndall test, in particular, to detect proteins or other particulate matter present in bodily fluids.
Pharmaceutical and food science industries use it for quality control and product development purposes. Its presence provides researchers with essential insight into colloidal suspension properties as they react when exposed to light.
Historical Background of Scattering of Light
- Ancient Observations: Light’s scattering has long been observed and documented since antiquity. Philosophers and scientists in ancient Greece, including Aristotle, made observations regarding sunlight’s propagation through materials such as water and air.
- Newton’s Discoveries: In the 17th century, Sir Isaac Newton conducted experiments using prisms, discovering that white light can be split up into its component colors using prisms. While he did not directly investigate scattering phenomena, his experiments laid down a strong foundation for understanding light’s behavior.
- Rayleigh Scattering: Lord Rayleigh, a British scientist in the 19th century, made significant contributions to our understanding of light scattering. His research focused on small particles or molecules scattering light in the atmosphere and explained why the sky looks blue during the day and when setting sun appears red or orange at sunset.
- Mie Scattering: At the turn of the 20th century, German physicist Gustav Mie built upon Rayleigh’s work by developing a theory to describe light scattering from larger particles or those possessing different optical properties – this work contributed greatly to our understanding of phenomena like sunset colors and halos formation.
- Modern Advancements: Light scattering research has advanced dramatically with technological developments in optics, materials science, particle characterization techniques, and computer simulation. Scientists now use sophisticated instruments and computer simulations to study various aspects of light scattering as applied to fields like atmospheric science, astronomy, and particle analysis.
What is Scattering of Light?
Scattering of light is an observable phenomenon that occurs when light interacts with matter, changing direction and dispersing in multiple directions. It results from interactions between photons of light and irregularities within its medium such as particles or irregularities within an environment where it passes.
When passing near particles or irregularities, light can absorb into them and be reemitted in different directions, diverging from its original path and altering its original trajectory.

There are two primary forms of light scattering, Rayleigh scattering and Mie scattering. Rayleigh scattering causes the blue color of the sky, occurring when particles that scatter light are much smaller than its wavelength.
Mie scattering occurs when scattering particles are comparable or larger than their wavelength. It results in phenomena like sunset colors, halos around sun, moon combinations, or light scattered by large particles or droplets in clouds.
The light scattering is vital in many scientific disciplines and daily experiences. Atmospheric science uses it to explain sky colors and the formation of rainbows; while astronomy uses it to interpret star and planet colors.
Particle characterization techniques use light scattering techniques for studying particles found within various materials. Overall, light scattering is an amazing phenomenon with profound ramifications across an array of fields.
Historical background of the Tyndall Effect
- Early Observations: Scientists had noticed the scattering of light for some time prior to John Tyndall’s work; however, without any clear explanation. Early observations included blue skies, laser beams visible through fog and colloidal suspensions having bluish hues.
- Tyndall’s Research: John Tyndall conducted experiments in the mid-19th century to study light scattering. Using a beam of light and various suspensions such as solutions of liquids with suspended particles called colloids and gases with fine dust particles, he observed that when light passed through these suspensions it scattered in all directions, making its path visible.
- Explanation: Tyndall explained that light scattering in colloidal suspensions was due to interaction between light and suspended particles, with shorter wavelengths such as blue and violet light being scattered more than longer wavelengths, giving rise to its characteristic blueish appearance.
- Scientific Impact: Tyndall’s work on the Tyndall Effect had a dramatic effect on optics, colloid chemistry and atmospheric science. His studies provided us with an explanation for why the sky appears blue while further expanding our knowledge about colloids and particulate matter found in various solutions.
- Legacy: John Tyndall’s research into the Tyndall Effect remains an integral component of optical science, with applications in fields as diverse as chemistry, medicine, and materials science. His contributions to light scattering continue to be studied and expanded upon by scientists today.
Difference Between Tyndall Effect and Scattering of Light
The Tyndall effect is a form of scattering light. The main difference in the Tyndall effect and light scattering is the fact that the Tyndall effect results from the result of scattering light caused by the particles that are in the path of the colloidal mix.
While the scattering of light is the scattering of light across all directions caused by the particles present along its route. Additionally, whereas the Tyndall effect is present in colloidal dispersions, such as suspensions, light scattering could occur in any environment that contains particles.
Here’s a concise comparison chart between the Tyndall Effect and the Scattering of Light in seven rows:
| Aspect | Tyndall Effect | Scattering of Light |
|---|---|---|
| Definition | Scattering of light by small particles in a medium, makes the path of light visible. | The general phenomenon is where light changes direction and disperses when interacting with matter. |
| Mediums | Liquids (colloidal suspensions), some gases, and transparent solids. | Gases, liquids, solids, biological tissues, and various media with particles. |
| Particle Size | Small particles or colloidal suspensions. | Varies depending on the medium, can include small and large particles or structures. |
| Wavelength Dependency | Generally less wavelength-dependent and more influenced by particle size and concentration. | Strongly wavelength-dependent, with shorter wavelengths scattering more effectively. |
| Mechanisms | Scattering due to small particles and colloids. | Diverse scattering mechanisms, including Rayleigh and Mie scattering, depend on the medium. |
| Examples | Milk, fog, mist, certain gemstones, underwater visibility. | Blue sky, sunsets, halos, glint in snow, cataracts, and various natural and artistic phenomena. |
| Practical Applications | Identifying colloids in solutions, diagnostic tests in medicine, and materials science. | Atmospheric science, astronomy, photography, materials science, medical diagnostics, and more. |
Exploring Light Scattering: How Does it Differ from the Tyndall Effect?
Definition of Light Scattering:
- Light Scattering: Light scattering refers to any phenomenon in which light interacts with matter and causes its path of travel to change or disperse in multiple directions, using various scattering mechanisms like Rayleigh and Mie scattering.
- Tyndall Effect: The Tyndall Effect is a type of light scattering. It occurs when light is scattered all directions by small particles or colloidal suspensions present within an opaque medium, thus rendering its path visible.
Scope of Light Scattering:
- Light Scattering: Light scattering encompasses an expansive set of phenomena which result in light being scattered by particles, molecules or irregularities in a medium, including atmospheric conditions, astronomy observations and particle characterization techniques.
- Tyndall Effect: The Tyndall Effect is a sub-category of light scattering that specifically refers to small particles or colloids scattering light.
Light Scattering Mechanisms:
- Light Scattering: Light scattering mechanisms vary greatly based on factors like particle size, composition and wavelength of light emitted by light sources. Some examples include Rayleigh scattering for smaller particles present in the atmosphere as well as Mie scattering (larger particles).
- Tyndall Effect: The Tyndall Effect is the scattering of light by small particles or colloidal suspensions; however, its exact mechanism remains unknown.
Wavelength Dependency in Light Scattering:
- Light Scattering: Light scattering can often depend strongly on wavelength. Shorter wavelengths (blue and violet) tend to scatter more, producing the blue hue seen in skies due to Rayleigh scattering.
- Tyndall Effect: The Tyndall Effect is typically less wavelength-dependent and more responsive to factors such as particle size and concentration.
Applications:
- Light Scattering: Light scattering can be found across many scientific disciplines, explaining things such as the blue hue of the sky, sunset colors and atmospheric phenomena as well as particle characterization techniques.
- Tyndall Effect: The Tyndall Effect is used primarily to identify colloids in solutions, conduct diagnostic tests in medicine, and understand the behavior of colloidal suspensions.
The Science Behind the Tyndall Effect: Why Does Light Scatter in Certain Substances?
- Size of Particles: Particle or molecular sizes in a medium are an essential consideration since when their diameter approaches that of light wavelength, these particles can effectively scatter it and lead to the Tyndall Effect being more pronounced in colloids with smaller particles.
- The wavelength of Light: The wavelength of light also plays a pivotal role. Shorter wavelengths like blue and violet light scatter more readily than longer ones like red or yellow light; this fundamental principle known as Rayleigh scattering applies here.
- Refractive Index: The refractive index of both particles and their surroundings determines how much light is scattered, with any significant discrepancies between their refractive indices amplifying this scattering effect.
- Density and Concentration: Particle density and concentration both play an integral part in determining the intensity of the Tyndall Effect. Higher particle concentrations cause more scattering, amplifying its impact.
- Path Length: Light’s path length through any medium also plays a significant role. Longer path lengths provide more opportunities for light to interact with particles, creating more noticeable Tyndall Effects.
- Wavelength of Light Source: The color of light sources used can influence the visibility of the Tyndall Effect. For instance, using blue laser pointers makes the Tyndall Effect more obvious.
Tyndall Effect and Light Scattering in Everyday Life
- Sunlight Filtering Through Trees: As sunlight filters through tree leaves, the Tyndall Effect becomes evident. Dust particles, pollen and water droplets present in the air scatter sunlight into visible beams of light that enhance their green appearance while creating visible beams of illumination from behind them.
- Headlights in Fog: Fog is composed of microscopic water droplets suspended in the air, so when headlights or streetlights shine through it, their light scatters all directions, diminishing visibility and creating a diffused, diffused appearance that reduces visibility – an effect which has important ramifications for road safety.
- Clouds and the Sky: Clouds consisting of water droplets or ice crystals scatter sunlight, giving them their characteristic appearance. Meanwhile, blue skies in daytime result from Rayleigh scattering due to shorter wavelengths being scattered by air molecules.
- Milk in Tea or Coffee: When adding milk to tea or coffee, it can produce an cloudy appearance due to light scattering off milk proteins and fat globules, known as the Tyndall Effect.
- Laser Light Shows: Laser light shows are often found at entertainment venues and involve the scattering of laser beams off airborne particles or smoke to produce mesmerizing visual effects that draw people in.
- Rainbows: Rainbows are breathtaking examples of light scattering. They occur when sunlight refracts off raindrops and disperses through them, eventually dissecting into its component colors as a rainbow forms.
- Camera Lens Flares: When it comes to photography, lens flares often result from light scattering within the camera lens itself and appear as streaks or spots of light that add visual interest and drama to photographs. They may add artistic or dramatic effects when added deliberately into images.
- Holograms: Holograms can be found everywhere from credit cards and product packaging to art installations, using light scattering technology to produce three-dimensional images which alter perspective as you move.
- LED Indicator Lights: Electronic devices such as smartphones and appliances feature small LED indicator lights which emit scattered light that makes their glow visible, sometimes in vibrant hues. This scattered illumination makes up one element of their illuminated casings.
- Fireflies: Bioluminescence in fireflies is an example of light scattering within the animal kingdom. Their bioluminescence emanates light in various directions, creating their unique glow.
Tyndall Effect and Scattering of Light in Different Mediums
The Tyndall Effect and light scattering are optical phenomena that manifest themselves differently depending on their medium of manifestation. Gases such as Earth’s atmosphere exhibit Rayleigh scattering which causes daytime skies to appear blue due to efficient scattering of shorter wavelengths by air molecules.
Tyndall Effect is most visible when light interacts with colloidal suspensions like milk or paint, where tiny particles scatter light and cause it to become opaque or turbid. Water bodies with suspended particles also display this phenomenon, as do gemstones with intricate structures or impurities that display it – creating unique visual effects such as color play or glint.
Scattering can significantly affect the optical properties of biological tissues like human eyes and skin. Protein aggregates in cataractous eyes may scatter light, leading to cataracts and vision impairment; collagen fibers and structures present on the skin also play a part in this phenomenon, altering perceptions of skin color and texture perceptions.
The Tyndall Effect and light scattering have an immense influence on our perceptions of reality, providing invaluable tools in scientific inquiry, art creation and day-to-day experiences.
Summary
The Tyndall Effect and Scattering of Light are optical phenomena involving light’s interaction with matter. More specifically, the Tyndall Effect refers to small particles or colloidal suspensions scattering light and making its path visible. Identification of colloids in solutions and their applications across medicine and chemistry.
On the other hand, light scattering refers to various mechanisms including Rayleigh and Mie scattering which occur throughout nature. Light interaction between matter and light occurs when light interacts with matter to alter its direction and dispersion, and is crucial in explaining sky color, atmospheric phenomena, astronomy and particle characterization.
While the Tyndall Effect is relatively wavelength-independent and typically restricted to small particles, the Scattering of Light encompasses various scattering mechanisms with wide-ranging applications in science and daily life.