Introduction of Isosteres and Bioisosteres
The major distinction between bioisosteres and isosteres is the fact that isosteres are compounds or ions having similar electronic and physical properties as well as shapes Bioisosteres, on the other hand, are bioactive compounds made up of an isostere.
Bioisosteres and Isosteres are two connected concepts. Isosteres could be defined as any ions or molecules that have the exact number of electrons in valence as well as having physical or chemical similarities.
Bioisosteres can be described as chemical substitutes or groups with identical physical or chemical characteristics with properties that are broadly similar in comparison to other chemical compounds. The two terms are typically utilized in the context of research and development in the field of bioactivity as well as drug development.
What are Isosters?
Isosters, in terms of chemistry, refer to compounds or functional groups with similar chemical and physical properties. Such isosteric compounds share key characteristics with each other such as having equal numbers of valence electrons as well as having comparable molecular volumes, bond lengths, and bond angles which give rise to similar chemical behavior.
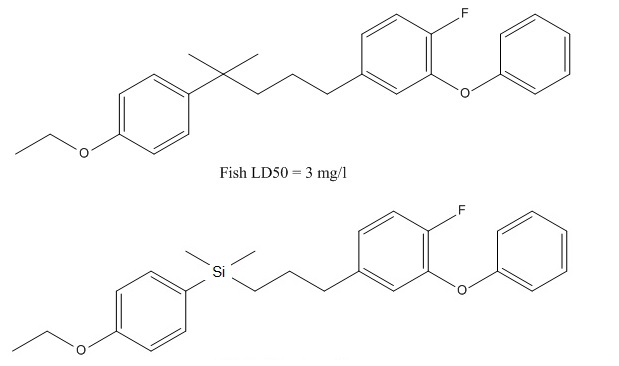
One outstanding example of isosterism can be seen with fluoride (F-) and hydride ions (H-). Although they come from distinct elements, both possess identical amounts of valence electrons; due to this similarity in size and charge, they often display comparable chemical reactivity.
Isosters play an essential role in medicinal chemistry and drug design. Medicinal chemists leverage isosteric pairs to strategically alter drug molecules while still meeting desired biological activity and pharmacological properties.
This may result in improved pharmacokinetics, reduced toxicity, and stability improvement as well as more potency pharmaceutical agents being produced by scientists through understanding and applying isosteric principles ultimately producing more therapeutic agents with improved therapeutic efficacy and greater patient benefits.
What are Bioisosteres?
Bioisosteres play an essential role in medicinal chemistry and drug design. They refer to compounds or functional groups with distinct chemical compositions or structures that share similar biological activities.
When substituted into molecules, bioisosteres can help preserve or enhance desired pharmacological effects while permitting structural modifications, unlike classical isosteres which prioritize chemical properties over biological interactions with receptors or enzymes.
Bioisosteres may involve switching out one hydrogen atom with a fluorine atom or replacing one functional group for another that still achieves similar biological results. These structural modifications may alter a drug’s pharmacokinetics, metabolic stability, and selectivity for target tissues while simultaneously maintaining efficacy.
Bioisosteres offer medicinal chemists an invaluable arsenal to optimize lead compounds, overcome drug development hurdles, and produce more effective and safer pharmaceutical agents.
When employed effectively bioisosteres can balance out therapeutic benefits with potential drawbacks thereby contributing to drug discovery and development progressing as a field.
The Role of Isosteres and Bioisosteres in Drug Design
Here is an overview of their roles in drug design:
Isosteres in Drug Design:
- Chemical Similarity: Isosteres are used to identify compounds or groups with similar chemical structures and properties, making their identification crucial when designing molecules with certain chemical behaviors.
- Structure-Activity Relationship (SAR) Studies: Isosteres provide medicinal chemists a valuable insight into the relationships between chemical structure and biological activity, with researchers being able to test how changing isosteres impacts the efficacy of drugs.
- Reactivity and Synthesis: Isosteres are used to alter a molecule’s reactivity or stability, so chemists often add them as isosteric substitutions either to improve its synthetic accessibility or fine-tune its properties.
- Patent Protection: Isosteres can be utilized strategically in drug design to create unique compounds not protected by existing patents and safeguard their intellectual property for pharmaceutical companies.
Bioisosteres for Drug Development:
Design Considerations:
- Optimizing Pharmacological Properties: Bioisosteres can be used to replace or modify parts of molecules to enhance their pharmacological properties such as increasing potency, selectivity for specific targets, or decreasing toxicity.
- Metabolism and Bioavailability: Bioisosteres can be utilized to address problems related to drug metabolism and bioavailability, specifically their metabolism and absorption by altering chemical structures to influence how drugs are utilized by their target organ. Chemists use substitution techniques such as these in their research in order to influence whether drugs are metabolized or absorbed in our bodies.
- Toxicity Reduction: Bioisosteres are used to replace toxic or undesirable functional groups with less dangerous alternatives, thus improving a drug’s safety profile and making its usage safer for use by patients.
- Solubility Enhancement: Modifying molecules with bioisosteres can increase solubility in biological fluids, making the drug more readily absorbed and distributed within the body.
- Resistance Management: Bioisosteres can aid in fighting drug resistance by altering their structure to stop or at least reduce pathogen-based resistance mechanisms from emerging.
- Side Effect Mitigation: Bioisosteres can help mitigate undesirable side effects while still offering their therapeutic advantages, for instance by replacing toxic groups with bioisosteric groups which will potentially lessen adverse reactions.
- Patent Extender: Pharmaceutical companies can extend a drug’s patent life with bioisosteric modifications by applying bioisosteric modifications that extend market exclusivity for extended market exclusivity.
How Isosteres and Bioisosteres Influence Molecular Structure and Activity
Isosteres:
- Chemical Similarity: Isosteres refer to compounds or functional groups that share similar chemical properties and structures; simply replacing one group with its isosteric equivalent will preserve overall chemical similarity.
- Structure-Activity Relationship (SAR): Isosteres are used to explore SAR by replacing functional groups or atoms within molecules to assess which structural components are essential for biological activity. This approach helps reveal any unexpected biological actions caused by certain changes.
- Reactivity and Stability: Isosteres can alter both the reactivity and stability of molecules. For instance, replacing an unstable group with a more robust isostere may prevent degradation in biological systems.
- Patent Protection: Isosteres can be added to molecules in order to produce novel compounds that differ significantly from existing patented medicines, thus helping safeguard intellectual property rights.
Bioisosteres for Optimizing Properties:
- Optimizing Pharmacological Properties: Bioisosteres can be used to enhance the pharmacological properties of drugs by substituting specific groups or atoms with others in an attempt to increase the affinity of target receptors with this compound and boost the potency and efficacy of treatments.
- Reducing Toxicity: Bioisosteres can replace toxic or metabolically reactive groups with less hazardous alternatives, decreasing adverse reactions risk while improving drug safety.
- Metabolism and Bioavailability: Bioisosteres can alter how quickly or slowly drugs are metabolized in the body, providing greater bioavailability or slowing metabolism, prolonging drug action. These modifications could improve bioavailability or slow metabolism resulting in extended effects from taking a medication.
- Solubility Enhancement: Chemists have learned how to utilize bioisosteres as solubilization agents to increase the solubility of drugs in biological fluids, making sure it can reach its destination more successfully.
- Resistance Management: Bioisosteres may help manage resistance by altering drug structures to bypass resistance mechanisms present in pathogens.
- Side Effect Mitigation: Bioisosteres can reduce or eliminate unwanted side effects while maintaining therapeutic activity. For example, replacing reactive groups with bioisosteric groups may decrease toxicity.
- Patent Extension: Bioisosteric modifications can extend a drug’s patent lifespan and prevent generic competition while lengthening market exclusivity.
Leveraging Isosteres and Bioisosteres for Drug Discovery
- Structure-Activity Relationship (SAR) Studies: Isosteres and bioisosteres are indispensable tools in structural activity relationship (SAR) studies. By substituting functional groups or atoms within molecules systematically, researchers can quickly and precisely pinpoint those structural elements essential to biological activity an understanding that informs new drug candidate design with enhanced properties.
- Pharmacological Enhancement: Bioisosteres can be used to tailor drug molecules in order to enhance their pharmacological properties, for instance replacing one functional group with bioisosteric groups can increase binding affinity at its target receptor and hence potency.
- Toxicity Mitigation: Bioisosteres can help mitigate drug candidates’ toxicity. By substituting toxic or metabolically reactive groups with more nontoxic counterparts, chemists can lower the risk of adverse side effects and minimize any possible risks to human health.
- Metabolism Optimization: Bioisosteres can alter a drug’s metabolic fate. Modifications can be made to slow metabolism down, increase bioavailability or change metabolic pathways to optimize its overall effectiveness.
- Solubility Enhancement: Isosteres and bioisosteres can help increase a drug’s solubility, helping it reach its target site more rapidly in the body.
- Patent Protection and Market Exclusivity: Implementing bioisosteric modifications or isosteres can result in novel compounds not covered by existing patents, providing pharmaceutical companies a way to safeguard intellectual property while keeping market exclusivity intact.
- Overcoming Resistance: Bioisosteres are effective ways of countering drug resistance. By altering drug structure to bypass resistance mechanisms in pathogens, their usefulness may increase.
- Minimizing Side Effects: Chemists have discovered bioisosteres to be useful tools in mitigating or eliminating undesirable side effects while maintaining the therapeutic activity of drugs containing bioisosteres. This strategy greatly increases patient safety and compliance.
- Structure-Based Drug Design (SBDD): Bioisosteres are used in SBDD to develop drug candidates based on three-dimensional structures of target proteins. This approach results in drugs that precisely meet binding sites on targets, increasing specificity and efficacy.
- Rational Drug Design: Isosteres and bioisosteres play an essential part in rational drug design. Medicinal chemists utilize their knowledge of chemical similarity and reactivity to craft molecules that interact optimally with biological targets, speeding up drug discovery.
Comparison Between Isosteres and Bioisosteres
Bioisosteres and isosteres are both related substances. One of the main differences between bioisosteres and isosteres is the fact that isosteres can be described as molecules or Ions with the same physical and electronic characteristics Bioisosteres, on the other hand, are bioactive compounds made up from an isostere.
A few examples of isosteres comprise SH CH3, NH2 and SH as well as aromatic rings like aminopyrine dienestrol and estradiol. are all examples of bioisosteres.
Here’s a comparison chart summarizing the key differences between isosteres and bioisosteres:
| Aspect | Isosteres | Bioisosteres |
|---|---|---|
| Definition | Chemical compounds/groups with similar structures and properties, especially with regard to valence electrons and molecular shape. | Chemical substitutions in a molecule that lead to similar biological activity, typically in drug design. |
| Context | Primarily used in chemistry to understand chemical similarity between compounds. | Applied in pharmaceuticals and biology for designing and optimizing drug candidates to interact with biological targets. |
| Purpose | Identify compounds with analogous chemical properties, facilitating chemical reactions or compound synthesis. | Modify molecules to maintain therapeutic effects while potentially improving safety, solubility, or other pharmacological properties. |
| Examples | Replacement of a carbon atom with a nitrogen atom (-CH3 to -NH2). | Substituting a functional group to maintain drug potency while addressing issues like toxicity or metabolism. |
| Applications | Chemical synthesis, structure-activity relationship (SAR) studies, understanding reactivity of compounds. | Drug design and optimization, addressing issues related to drug toxicity, metabolism, and selectivity for the target biomolecule. |
What is the similarity of Isosteres and Bioisosteres
Here are the main similarities between isosteres and bioisosteres:
- Chemical Substitutions: Isosteres and bioisosteres involve replacing certain atoms, functional groups or structural elements within molecules in order to form new forms that differ chemically and structurally from its parent compound.
- Modifying Molar Structure: As with both cases, this project seeks to change the molecular structure of an organic compound while keeping or improving certain properties intact.
- Enhancement of Specific Properties: Isosteres and bioisosteres can be used to increase certain characteristics of molecules, including their biological activity, solubility, metabolic stability or safety.
- Drug Design and Optimization: Both concepts are applied when designing and optimizing drugs to create more effective and safer medications.
- Chemical Similarity: Isosteres and bioisosteres rely heavily on chemical similarity for their activities. Isosteres utilize this property in order to preserve or modify chemical properties while bioisosteres use similarity for some pharmaceutical benefits.
Summary
Isosteres and bioisosteres are fundamental concepts in both chemistry and drug design. An isostere is defined as two or more molecules with similar chemical and physical properties that share similarities. Its use allows medicinal chemists to modify drug molecules while keeping desired characteristics unchanged.
Bioisosteres, on the other hand, are compounds or functional groups with distinct chemical structures but equivalent biological activities when present within molecules. Bioisosteres play an integral part in drug development, specifically optimizing pharmacological interactions, increasing drug safety, and improving pharmacokinetics.
While isosteres focus more on maintaining or increasing chemical properties than maintaining or increasing biological activity contributing significantly towards designing effective yet safe pharmaceutical agents.