Natural and Artificial Transmutation: Transmutation refers to the alteration of the atomic nuclei leading to the transformation from a chemical component to an entirely different chemical element. There are two kinds of transmutation: artificial and natural transmutation. The major difference between artificial and natural transmutation is that natural transmutation involves the radioactive decay which occurs in the core of stars, whereas artificial transmutation involves the conversion of an element into a different element in a controlled manner.
Definition of Transmutation
Transmutation is the process of changing or transforming one element into another one by altering the number of protons that are present in the nucleus of the atom. This process can happen in two primary ways: natural transmutation which occurs spontaneously in some radioactive elements, and also artificial transmutation, in which you induce the transformation by external means like neutron capture or particle bombardment.
Transmutation is a concept that is a key element in many fields of science such as chemical physics, nuclear science, and medicine. It has resulted in significant advances in understanding the structure of atoms and in the development of new isotopes for diverse applications.
Importance and applications of transmutation in various fields
Transmutation is a key factor in many scientific disciplines. It has been able to discover numerous applications that have a profound impact on human technology, knowledge as well as medicine. Here are a few most important areas in which transmutation plays an important role:
Nuclear Physics and Chemistry:
Transmutation is a key element in studying nuclear physics as well as the study of chemistry. It assists researchers in understanding the behavior of nuclei in atomic form and their stability as well as the mechanisms that lead to radioactive decay. Transmutation research has resulted in the discovery of a variety of nuclear reactions, as well as the development of nuclear models, which contribute towards our comprehension of fundamental forces that control matter.
Nuclear Energy and Power Generation:
Artificial transmutation plays a crucial part in nuclear energy production. Through processes such as the nuclear fission process, heavier metals can be bombarded with neutrons in order to trigger a chain reaction, producing a massive quantity of power. A controlled nuclear reaction forms the foundation of the nuclear power plant, which provides an effective and reliable source of energy without releasing greenhouse gases.
Radioisotope Production:
Artificial transmutation can be used to create a range of radioisotopes that are used in a variety of areas, including industry, medicine, and agriculture. The radioisotopes are used for medical imaging, chemotherapy treatment cleaning medical devices and tracking the movement of substances within biological systems.
Nuclear Medicine:
Transmutation has revolutionized the field of medical diagnostics and treatment using nuclear medicine. Radioactive isotopes generated through artificial transmutation are utilized in procedures like positron emission tomography (PET) and single-photon emission computed tomography (SPECT) to detect and detect ailments. Radiotherapy targeted, commonly referred to as radioisotope therapy, can be utilized to treat specific types of cancer.
Material Science:
Artificial transmutation plays an important role in changing the properties of the material for specific uses. By bombarding the material with high-energy particles can alter their structure and atomic structure and make them better suited to various scientific and industrial applications.
Astrophysics and Cosmology:
Natural transformation processes, like nucleosynthesis in stars, are crucial in determining the chemical composition of the universe. Stars’ fusion reactions create elements that range from hydrogen and helium to heavier elements such as carbon oxygen and iron. This process has significantly affected the variety of elements that we see in the universe.
Environmental Remediation:
Transmutation methods are being studied as potential solutions to the management of nuclear waste and environmental remediation. Through the transmutation of radioactive isotopes with long-lived lives into more stable, shorter-lived elements, scientists seek to lessen the environmental impacts and the risks that come with the disposal of nuclear waste.
Transmutation is an essential process that has far-reaching implications. The applications of nuclear energy production, physics medicine, material science, and astrophysics have dramatically improved our knowledge of our universe. They have also allowed the development of technology that helps humanity in a variety of ways.
The distinction between Natural and Artificial Transmutation
The main difference between artificial and natural transmutation is in their source as well as the method of transformation. Let’s examine the major differences between the two types of transformation:
Origin of the Transmutation Process:
- Natural Transmutation: The natural transmutation takes place naturally in radioactive elements that are found in nature. They undergo radioactive decay because of the unstable nature of their nuclei atomic. As a result, they release radiation or particles and transform into various elements with stronger nuclei.
- Artificial Transmutation: Artificial transmutation in contrast is the process of inducing nuclear transformations within elements that don’t necessarily go through spontaneous decay. It requires external methods for example neutron capture or particle bombardment to start the process of transmutation.
Occurrence:
- Natural Transformation: Natural processes of transmutation occur continuously throughout the world as well as on Earth and in the universe. Radiative decay, as well as cosmic rays, can be two examples of natural transmutation processes.
- Artificial Transmutation: Artificial Transmutation is an artificially-created process that generally occurs in controlled environments such as labs or facilities for nuclear. It requires sophisticated equipment like particle accelerators and nuclear reactors to produce the required conditions to allow transmutation.
Rate and Controllability:
- Natural Transmutation(Natural): The natural transmutation procedures have probabilistic decay rates and clearly defined half-lives. The decay rate is unique to each radioactive element, and can not be controlled or changed by means external to the element.
- Artificial Transmutation (AUT): This technique enables to control the speed in nuclear reactions. The energy and intensity of the neutron or particle beam source that is used can be altered to affect the rate of transmutation reactions.
Types of Elements Involved:
- Natural Transformation: Nature transmutation typically involves radioactive elements and isotopes that naturally degrade in time. They are typically present in small amounts within the Earth’s crust and are present since the creation in the Universe.
- Artificial Transmutation: Artificial transmutation may be a result of unstable or stable elements. Stable elements can transform into radioactive isotopes by bombarding them with neutrons or particles. In contrast, radioactive isotopes may be converted to stable elements by specific transformation processes.
Applications and Significance:
- Natural Transmutation Processes: Natural transmutation procedures are essential to understanding the behavior that radioactive substances exhibit, the determination of the oldest materials by radiometric dating, as well as the creation of certain cosmogenic elements that are used in archaeology and geology.
- Artificial Transmutation: Artificial transmutation can be used for a variety of purposes. It is employed to produce radioisotopes to aid in cancer treatment, medical imaging and industrial applications. In addition, artificial transmutation is vital to the nuclear energy industry as well as research in material science as well as environmental cleanup.
The difference between artificial and natural transmutation is in their spontaneity appearance in specific elements, and the controlled induction through nuclear subatomic transformations. Their speed and the controllability of the process and the type of components involved. Both artificial and natural transmutation have important technological, scientific and medical impacts, enhancing our understanding of the fundamental processes in atomic physics and providing a variety of practical applications.
Natural Transmutation
The natural transmutation process, sometimes referred to as spontaneous transmutation, is also known as radioactive decay. It is a process whereby unstable elements are transformed into various elements in order to reach improved stability. It happens without external intervention and is driven by the intrinsic instability of nuclear nuclei of certain isotopes.
The most important characteristics of natural transmutation:
Natural transmutation involves radioactive isotopes which are elements that have unstable nuclei. They contain too many neutrons, or too few, in relation to the quantity of protons, which makes their nuclei unstable.
- Decay Modules: Radioactive elements can decay in a variety of ways which are all related to the emission of particular radioactive particles. The most popular decay types comprise beta decay, alpha decay (both beta-minus and beta-plus) Gamma decay, and spontaneous fission.
- Alpha Decay: During alpha decay, an unstable nucleus produces an alpha particle that is a helium nucleus comprised of two protons and two neutrons. The result is that the atomic number decreases by two and the mass number by four.
- Beta decay: Beta decay refers to the release of beta particles which can be electrons (beta-minus decay) or positrons (beta-plus decay). Beta-minus decay boosts the number of atomic nuclei by one, whereas beta-plus decay reduces its number by one.
- Gamma Decay: After alpha or beta decay the daughter nucleus might remain in an exuberant state. The nucleus releases energy that is in excess the gamma radiation in order to move into a more stable state.

- SPONTANEOUS FISSION: in this form, heavy nuclei break up into two smaller nuclei, along with emissions of neutrons as well as other particles.
- Decay Rate and Half-Life: Each radioactive element has a characteristic decay rate that is expressed as a half-life. The half-life is the period it takes for an initial quantity of a radioactive element to go through the process of decay. It could range from fractions of a second up to billions of years, based on the elemental isotope.
- Importance: Natural transmutation has vital roles in various technological and scientific aspects. It is used to determine radiometric dates, which is a method of determining the age of artifacts and materials. Research into natural transformation processes can also help us gain an understanding of astrophysics, nuclear physics, and the behavior of elements within the surroundings.
- Cosmic Rays: Certain natural transmutations result from the collision of cosmic rays with high energy from space colliding with the atomic nuclei within the Earth’s atmosphere as well as the material that is on its surface. These interactions may result in the creation of new isotopes by spallation or various nuclear reactions.
The overall picture is that natural transmutation is a fundamental process that takes place all over the universe, affecting the number of elements in the universe and assisting in the formation of stars, and offering important insights into the behavior of nuclei in atomic form and the development of our planet.
Artificial Transmutation
Artificial transmutation also referred to as induced transmutation is a type of nuclear reaction in which unstable or stable isotopes of elemental elements change into a variety of isotopes, or even different elements using external sources. Contrary to natural transmutation that happens spontaneously the artificial process requires human intervention as well as the use of different methods and tools to trigger and regulate the nuclear transformations.
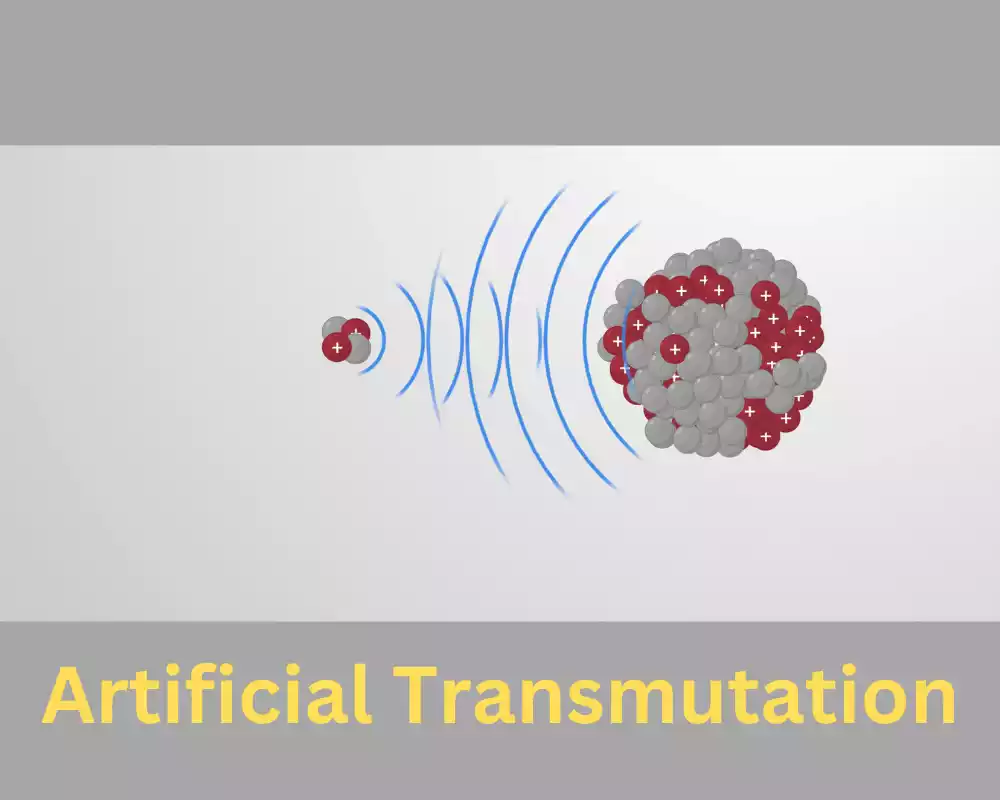
The most important features of synthetic transmutation:
- Particle Bombardment: One of the main methods employed in artificial transmutation is bombarding unstable or stable targets using high-energy particles. Particle accelerators, like linear accelerators and cyclotrons, are typically used to boost protons, neutrons, and other particles charged to extremely high speeds. The accelerated particles are targeted at the nucleus which triggers nuclear reactions.
- Neutron Bombardment: Neutron irradiation is a second method employed for artificial transmutation. Neutrons are neutral particles that are able to penetrate the nuclei of atoms without being pulled back through electrostatic force. When neutrons are taken up by the nucleus, they may trigger nuclear reactions, which can lead to the formation of new elements or isotopes.
- Production of Isotopes: One of the main reasons for artificial transmutation is to create of certain isotopes which aren’t naturally abundant or easily accessible. Certain isotopes that are used in nuclear medicine to aid in imaging and chemotherapy as well as for research and industrial applications can be manufactured in a laboratory setting through transmutation processes.
- Nuclear Reactors: Nuclear Reactors can be utilized to create controlled nuclear reactions, generating an array of isotopes as well as substances as byproducts. In certain instances transmutation reactions in nuclear reactors may be utilized to convert radioactive waste with a long life into isotopes with shorter lives, thus reducing the environmental impacts of storage of nuclear waste.
- Application: Artificial Transmutation can be used in numerous practical applications in a variety of areas:
- Nuclear Medicine: The use of radioisotopes produced by artificial means are utilized in medical imaging and scanning for diagnostic purposes (e.g. PET, PET or SPECT scans) and targeted radiation therapy to treat cancer.
- Isotope Production: Artificial transmutation is vital for producing radioisotopes used in radiopharmaceuticals, industrial tracers, and other applications.
- Nuclear Energy: Nuclear Energy in nuclear power stations, artificial transmutation via nuclear fission is utilized to produce energy.
- Material Science: Scientists may make use of artificial transmutation to alter the properties of the material and examine how nuclear interaction affects various materials.
- Control and safety: One of the major positives of using artificial transmutation is its capability to regulate and control the process. The energy and intensity of the beams of particles or neutron sources are modified to alter the rate and the outcome of transmutation reactions, which ensures security and precision.
Artificial transmutation is an effective tool that has helped us understand the complexities of nuclear physics. It has also facilitated the creation of vital isotopes used in a variety of fields and helped to make advancements in industry, medicine, as well as scientific research. It remains an effective method of advancing technology and knowledge of humans.
Burstiness in Transmutation Descriptions
Transmutation descriptions that are bursting with energy refer to the variety of sentence lengths and complexity in the text. It is the result of using a mixture of simple, short sentences with more complicated, longer sentences to produce a lively and captivating narrative. In addition, by incorporating a burst of humor it makes the text more appealing and interesting to readers, mimicking the way people naturally talk about complex subjects such as transmutation.
Examples of a burst in transmutation descriptions:
“Natural transmutation processes occur spontaneously in certain radioactive elements. These elements have unstable nuclei. As a result, they undergo radioactive decay. During this process, they emit particles or radiation. These emissions transform the elements into different isotopes or elements that are more stable.
One common form of natural transmutation is alpha decay. In alpha decay, an unstable nucleus emits an alpha particle. The alpha particle is composed of two protons and two neutrons, essentially a helium nucleus. This emission reduces the atomic number of the element by two and the mass number by four.”
Take note of how the paragraph above is a mix of shorter sentences as well as more lengthy, detailed ones. This creates an element of rhythm and flow the text and makes it simpler for readers to understand while keeping their attention. It also helps to draw attention to key aspects and make the text more memorable.
Through the use of snark in transmutation explanations, writers can effectively communicate complicated ideas in science and technology in a manner that is informative as well as enjoyable. It brings a personal touch to the information which makes it more relatable and enjoyable for the reader.
Perplexity in Transmutation Explanations
Transmutation explanations that are a bit confusing refer to the complexity of technical terminology used to describe the complex process of transmutation. It is the process of using specific vocabulary and terms in science to give a deeper and more thorough knowledge of the subject. Incorporating perplexity into the information is more clear and is suitable for those who have a solid background in the field of science in which they are interested.
Transmutation example of perplexity explanations:
“Natural transmutation, a stochastic phenomenon driven by the inherent instability of certain radioactive isotopes, instigates spontaneous nuclear transformations wherein these isotopes undergo decay processes, emitting alpha, beta, or gamma particles and ultimately transmuting into new isotopes or even different elements with more stable atomic nuclei. The rates of these decay processes are dictated by their respective half-lives, and intrinsic properties of the isotopes in question, and are characterized by probabilistic fluctuations.
Alpha decay involves the ejection of alpha particles, comprising two protons and two neutrons, resulting in a decrease of two units in the atomic number and four units in the mass number of the parent nucleus.”
In this case, the use of scientific terms, like “stochastic phenomenon,” “decay processes,” “half-lives,” “probabilistic fluctuations,” and “atomic number” and “mass number,” is a source of confusion in the understanding. These terms offer an accurate and complete understanding of the processes in transmutation, which makes the information appropriate for those who are familiar with nuclear physical sciences and related areas.
Through the inclusion of perplexity in explanations of transmutation, authors can appeal to a more targeted audience, including researchers, scientists and students who are studying nuclear physics, Chemistry, and other fields that deal with transmutation. This allows for an in-depth exploration of the subject and promotes the critical thinking and analysis of those who have a good understanding of the subject.
Conclusion
Transmutation refers to the alteration of the nuclei in atomic atoms which leads to the transformation of a chemical element to another chemical element. There are two forms of transmutation, natural and artificial transmutation. One of the main differences between transmutations that are natural and artificial is that natural transmutation occurs through the decay of radioactivity that happens at the heart of stars and artificial transmutation refers to the process of converting an element to another.

