Dispersion and Diffusion
Dispersion and Diffusion are both processes that involve the spreading or scattering of particles or substances, but dispersion typically refers to the separation or scattering of different components within a medium, while diffusion refers to the gradual movement of particles from an area of high concentration to an area of low concentration.
Dispersion is the spreading or scattering of an object or information. It may occur because of causes like mixing, or changes in the data’s values. The terms diffusion and dispersion are separate terms with distinct definitions and uses. Dispersion relates to the spread or scattering of an object or information. In contrast, diffusion is specifically related to the motion of particles from areas with higher concentrations toward an area with a lower concentration.
Dispersion phenomenon is observed in many diverse scientific contexts, including physical sciences or chemistry. It can also be seen in statistics. It is the spread of or mixing of substances as well as the dispersion of data elements. The cause of dispersion could be elements like mixing, or fluctuations in the data’s values. It is usually measured with statistics such as variabilities, ranges, and standard deviation.
The majority of diffusion occurs in chemistry, physics, and biology. It’s a process that involves particles moving between areas with higher concentrations to areas of smaller concentrations, powered by gradients of concentration. It can occur in gas and solutions as well as in biological systems. The speed of diffusion can be dependent on factors like temperature, concentration gradient as well as molecular size, and shape.
The distinction between diffusion and dispersion is essential in the context of science in order to describe accurately and analyze methods that deal with the spread or movement of substances, or information.
What is Dispersion
Dispersion is the term used to describe the spread or scattering of any substance or information. In physics, the term dispersion usually is a term used to describe the process by which light is split into component hues when it passes through a prism or media with different refractive indices. The reason for this is that various wavelengths of light move at different speeds, leading to the equilibration of hues.
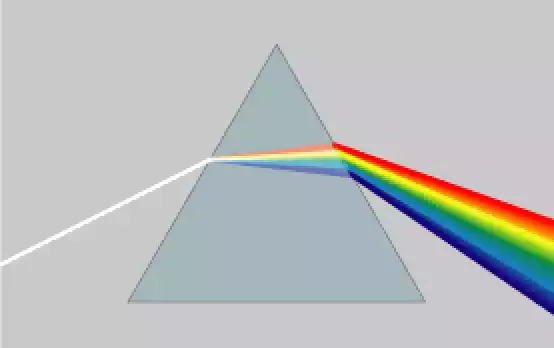
In the field of chemistry, dispersion may be defined as the spreading or mixing of different substances in a. In the case of an amount of dye placed in water in a glass, dye molecules break up, and spread across the entire water until they’re uniformly dispersed. The measures of dispersion including variabilities, ranges, and standard deviation, give an insight into how points are distributed in relation to the central trend which is the mean or median.
Understanding the concept of dispersion is crucial for a variety of scientific research. It assists in understanding the properties of light, studying the interaction of different substances as well as assessing the variation or the spread of information points used during statistical analysis.
What is Diffusion
Diffusion describes the way in that molecules or particles move across an area with a higher concentration to another area with a lesser concentration. It is an essential notion in physics, chemicals as well as biology.
In the field of physics, diffusion happens as gas molecules move around and mix without restriction because of their energy kinetics. This causes the dispersal of gas molecules in a given area until they are equally dispersed.
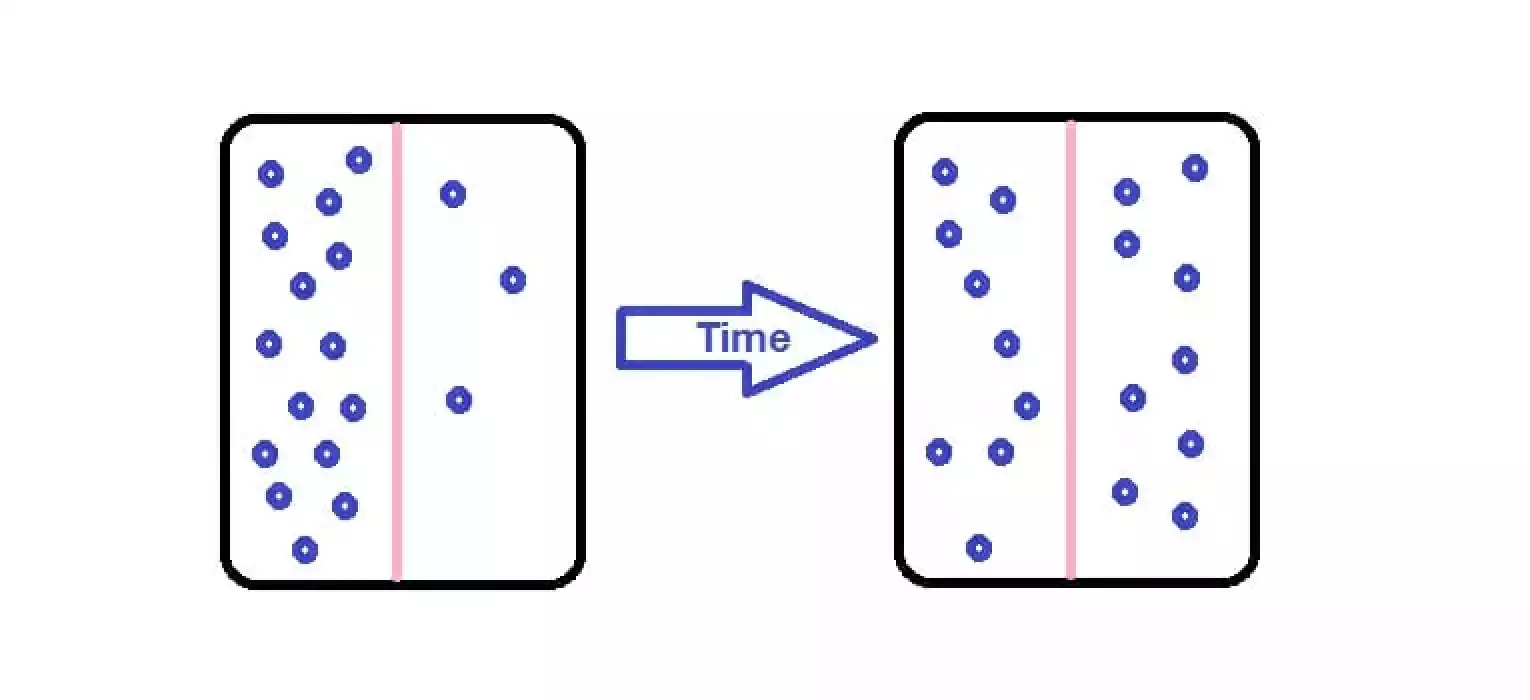
In the field of chemistry, diffusion can be evident when particles of solute disperse and circulate through the solvent, leading to the creation of a homogeneous mixture. The speed at which diffusion occurs is contingent on various factors, including the gradient of concentration along with the temperature as well as the dimensions and shapes of the molecules that are involved.
In the field of biology, diffusion plays a vital role in cellular processes. As an example, nutrients as well as substances that are wastes diffuse through cells’ membranes in order to ensure homeostasis inside cells. Carbon dioxide and oxygen are absorbed through the respiratory system as respiration occurs.
Diffusion is caused by the inherent tendency of particles to migrate across areas of greater concentrations to regions with a smaller concentration, in the pursuit of the equilibrium. It is a crucial procedure for mixing, transporting, and exchange of chemicals across different systems. Understanding diffusion is essential for fields like biology, physics, chemistry as well as engineering. It can help explain things like gas exchange within the lungs, and the behavior of chemicals in solutions, as well as the motion of particles within physical systems.
Difference between Dispersion and Diffusion
Diffusion and dispersion are two different concepts that have different definitions and their applications.
Below are the major distinctions between diffusion and dispersion:
Definition:
- Dispersion: Dispersion means the scattering or spreading of a data item or substance. It is the way in which a material or data is spread or scattered.
- Diffusion: Diffusion is a method by which particles or molecules travel across an area with a greater concentration to an area that is lower in concentration. This is specifically referring to the motion of particles caused by concentration gradients.
Nature of Phenomenon
- Dispersion: Dispersion refers to the scattering or spreading of data or substances. It could be due to different factors, such as mixing or fluctuations in the data’s values.
- Diffusion: Diffusion is a result of the nature-given tendency of particles to migrate between regions with higher concentrations into areas of less concentration until they reach equilibrium.
Scope of Application
- Dispersion: It is an idea that can be applied to many sciences like the sciences of chemistry, physics, and statistics. The term can refer to things like the dispersion of light, the mixing of different substances and the spreading of information points.
- Diffusion: It is found in physics, chemistry as well as biology. It is the basis for processes like gas molecules moving and the dispersal of solvent particles inside a solvent or the exchange of compounds between cell membranes.
Measurement:
- Dispersion: Dispersion is assessed using measures of statistical significance such as variation, range and standard deviation. These measures can provide insight into the spread or dynamic range of the data points.
- Diffusion: It is usually monitored or recorded by monitoring the concentration changes or movements of particles. This is usually done with the help of mathematical models.
Focus:
- Dispersion: The term “dispersion” is usually related to variability or spread. It indicates how things or data points are scattered or dispersed.
- Diffusion: Diffusion is the spread or intermixing of particles owing to gradients in concentration, focusing on the transfer of particles from zones of greater concentration to those with lower concentration.
Knowing the distinction between diffusion and dispersion is crucial in the context of science because it permits accurate explanation and evaluation of procedures that involve the spreading of particles, scattering or the movement of data or substances.
Applications of Dispersion and Diffusion
Here are some applications of dispersion and diffusion:
- Environmental Monitoring: Dispersion and diffusion are crucial in studying the movement and dispersion of pollutants through the atmosphere, water, and in soil. Knowing the patterns of dispersion can aid in assessing and monitoring environmental pollution including the spreading of pollutants in the air resulting from industrial emissions, or the dispersion of pollutants in the water bodies.
- Dispersion techniques for pharmaceuticals: Formulations are commonly employed in formulations for pharmaceuticals to increase the dissolution rate and bioavailability of drugs. In dispersing active components into smaller pieces or increasing their absorption, medications will be more efficiently absorbed by the body, resulting in better outcomes for therapeutic purposes.
- Paints and coatings: Dispersion techniques are used for the creation of coatings and paints for the uniform distribution of pigments as well as other additives. A proper dispersion process ensures quality, gloss, and the quality of the coating while making sure that there is no settling or agglomeration of particles.
- Food and Beverage Industry: Dispersion and diffusion have a significant role to play to a variety of aspects of the industry of food and beverages. In the case of food production, dispersion can be employed to produce solid emulsions, suspensions, and foams. The role of diffusion is in distributing flavor throughout food items during cooking or process that results in delicious flavors.
- Environmental Remediation: The processes of diffusion are used in various environmental cleanups methods, like extraction of soil vapor and groundwater treatment. Contaminants move across high-concentration zones and low-concentration zones, which allows the removal of or treatment.
- Membrane Technology: Diffusion is a key component to membrane-related processes like ultrafiltration, reverse osmosis as well as gas separation. Through the use of concentration gradients membranes allow specific species to spread across the membrane, which allows the purification of water, separation of the components, as well as gas filtering.
- Energy Storage: The phenomena of diffusion and dispersion play a significant role in the field of energy storage. For instance, in lithium-ion batteries dispersion of active electrode materials as well as diffusion of lithium ions are key in the performance as well as energy capacity of the battery.
- Material Science: Dispersion, as well as diffusion, are crucial considerations in the field of material science. achieving uniform dispersion for particles or additives in the matrix could enhance the property of the material, for example, the strength, conductivity, and optical qualities.
- Chemical Reactions and Catalysis: Diffusion plays an essential role in catalytic and chemical reaction processes. By facilitating it to allow the dispersion of reactions as well as substances within a reaction process or over catalyst surfaces it allows efficient mass transfer as well as can influence the rates of reactions as well as selectivity.
- Pharmaceutical Inhalers: Dispersion and diffusion concepts are used in the development of inhalers for pharmaceutical use. The dispersion of medications into small particles permits an efficient and effective route to the lungs. This ensures effective absorption as well as therapeutic effectiveness.
Challenges and Advances in Studying Dispersion and Diffusion
These are a few issues and advancements in understanding diffusion and dispersion:
Challenges:
- Complexity of System: Researching dispersion and distribution in complicated systems such as multiphase flow or heterogeneous media is challenging due to the variety of interactions and different transport properties associated with them.
- Experimental Techniques for Investigating: Diffusion and dispersion often lack resolution in temporal and spatial dimensions in particular when working with the nanoscale and rapid process.
- Multiscale Phenomena: Dispersion as well as diffusion can occur on a variety of lengths and time scales. This makes it difficult to construct complete models that can are accurate in capturing the phenomena over a variety of dimensions.
- Interparticle interactions: Identifying and quantifying the impacts of interparticle interactions including agglomeration and adhesion, and surface interactions on diffusion and dispersion behavior are difficult because of the nature of these interactions.
- The Numerical Simulation: The complexity of Simulating Dispersion as well as diffusion using computer-based methods are computationally demanding and requires advanced models to take into account a range of elements that influence transport behavior.
Advances:
- Advanced Characterization Methods: The advancement of advanced techniques for characterization including high-resolution imaging and spectroscopy and particle tracking techniques can allow greater precision and precise examination of diffusion and dispersion processes on the micro- and nano-scale.
- Computational Modeling and Simulating: Modern technological advances in computational modeling, and simulation methods, which involve the use of complex algorithms as well as supercomputers, as well as multi-scale modeling methods that permit accurate calculations and simulations of diffusion and dispersion for complex systems.
- Nanotechnology as well as Materials Engineering: The advent of nanotechnology provides the possibility of control and manipulation of dispersion and diffusion processes on a nanoscale. This allows the creation of materials with personalized features and better dispersion properties.
- Machine Learning and Data Analytics: Data Analytics and Machine Learning: The use of machine-learning algorithms and analytical techniques for data to study massive simulation and experimental datasets has the capacity to reveal hidden patterns and relationships and improve our understanding of the processes of diffusion and dispersion.
- Advancement in Measurement and Visualization: Techniques Advanced methods of measurement and visualization like advanced microscopy techniques, real-time imaging as well as 3D visualization techniques allow researchers to examine and analyze dispersion and diffuse phenomena in real-time and with more sophisticated systems.
- Interdisciplinarity: Collaborating with researchers across disciplines like engineering, physics, chemistry and biology. It encourages the exchange of ideas techniques, methods, and strategies that lead to a greater understanding of diffusion and dispersion phenomenon.
The similarity of Dispersion and Diffusion
Dispersion and diffuse, despite their differing definitions and application, have some commonalities. The reason for this is their roles in the spread and scattering of materials or information. They differ on the basis of particular mechanisms and settings they are both related to dispersion. are key concepts across a range of science disciplines?
A commonality between dispersion as well as diffusion is the notion of spreading. When dispersion occurs, the particles or data can be scattered or dispersed which results in a wide spread. In the same way, diffusion can lead to the spread of particles as they move between regions with higher concentrations into areas with lower concentrations. Both times, the particles or the substances are much more evenly distributed throughout the entire system.
A further similarity is the importance of dispersion as well as diffusion across a range of scientific disciplines. The phenomenon of dispersion is seen in physics, chemistry as well as statistics. The concept of diffusion is used in the fields of physics, chemistry and chemistry as well as in biology. Multidisciplinary aspects of these concepts underscore the importance of both concepts to understand the chemical behavior of diverse chemical compounds and the process they play in.
Dispersion as well as diffusion are both involved in mixing substances. Dispersion is when chemicals are blended or mixed and result in a homogeneous distribution. In the same way, diffusion aids mixing by encouraging the transfer of particles across areas with higher concentration into areas with less concentration. This results in the intermixing of different substances as well as the creation of equilibrium.
In addition, dispersion and diffusion are affected by gradients in concentration. When dispersion occurs, the distribution of materials or data can be affected by fluctuations in concentration leading to different amounts of spread. The process of diffusion, on the other hand, is dependent on the gradient of concentration to be the main driver for the movement of particles. The existence of concentration gradients determines the direction of and speed of diffusion. The particles naturally moving from areas of more concentration to areas of smaller concentration.
Dispersion and diffusion can be involved in processes that require scattering or spreading. Dispersion is evident in phenomena like light dispersion in which light is split into components and then scatters away, creating the effect of rainbows. The phenomenon of diffusion is associated with a spreading phenomenon, for instance, the diffusion of gas molecules or the dispersal of the solute within a solvent. Both of the substance involved is subject to diffusion or scattering across the medium.
Diffusion and dispersion can have an impact on the analyses of data. Dispersion is the study of measures such as variabilities, ranges, and standard deviation can provide insight into the spread and variation of the data. The same way, the processes of diffusion can be studied and measured to understand the way particles spread or disperse throughout a set of. The understanding of dispersion and diffusion are crucial to understanding and accurately analyzing the data set.
Dispersion and diffusion share a number of similarities even though they differ in their ways of working and in their environments. Spreading concepts as well as multidisciplinary importance, the involvement in mixing processes, the influence on concentration gradients, involvement in the spreading process, and influence on the analysis of data are common elements of dispersion and diffusion. Understanding these commonalities enhances our understanding of the primary concepts that govern the behavior of chemicals as well as processes across a variety of research settings.
Summary
Dispersion is the spreading or scattering of an object or information. It may occur because of causes like mixing, or changes in the data’s values. The phenomenon of dispersion can be observed in science (light dispersion) as well as in the chemistry field (mixing of chemicals) and in statistical analysis (spread of information). It’s usually associated with the spread or variability of data and can be analyzed using statistics like variance, range, and standard deviation.
Diffusion On the other hand is the process of moving of particles from regions of greater concentration to those with less concentration. It’s controlled by concentration gradients and is a part of the sciences of physics (gas diffusion) and chemical processes (solution mixing) and biology (cellular process)

