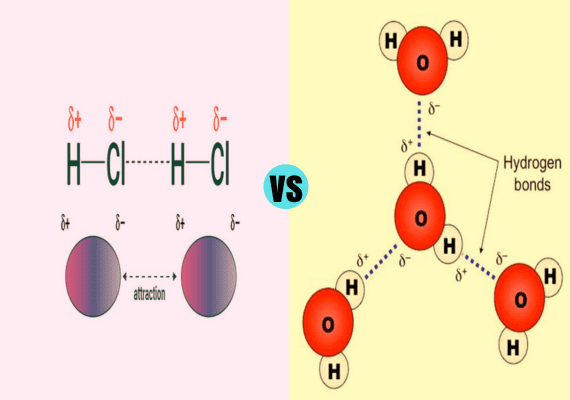
Dipole-Dipole Interactions and Hydrogen Bonding
Dipole Dipole Interactions and Hydrogen Bonding fact that dipole-dipole interaction occurs in the event that partially negatively charged molecules are in contact with another one with a positive charge while hydrogen bonding is one of the special types of dipole bonding that occurs between the O, and N as well as F and H hydrogen atoms.
Hydrogen bonds as well as dipole-dipole interactions are significant and are studied in molecular bonding research and molecular behavior crystal structure, as well as various other chemical aspects.
What are Dipole Dipole Interactions?
Dipole-dipole interactions are a type of intermolecular force found between polar molecules. This force results from unequal sharing of electrons among covalent bonds, leading to partial positive and partial negative charges within molecules.

Polar molecules exhibit strong electron attraction between their constituent atoms, with more electronegative ones drawing electrons more strongly, leading to them carrying partial negative charges (d-). This creates what’s called the molecular dipole moment – an abstract vector quantity representing how overall charges are distributed within a molecule.
Dipole-dipole interactions occur when one polar molecule’s positively charged end (d+) attracts to an oppositely charged end of another polar molecule (d-). This electrostatic attraction between oppositely charged ends of neighboring molecules results in relatively strong forces when compared with other intermolecular forces such as London dispersion forces.
Dipole-dipole interactions play a pivotal role in the properties of polar compounds, from their increased boiling and melting points compared to nonpolar ones, to higher surface tensions and greater solubility in polar solvents. Dipole-dipole interactions play a significant role in many areas of chemistry, biology, and everyday life.
What is Hydrogen Bonding?
Hydrogen bonding is an intermolecular force that occurs between molecules when hydrogen forms covalent bonds to electronegative elements such as oxygen, nitrogen or fluorine atoms. Hydrogen bonding occurs when an electronegative hydrogen atom, partially charged due to its electronegativity, forms an attractive interaction with another electronegative atom in another molecule.
Hydrogen bonds feature an intense electrostatic attraction, making them significantly stronger than regular dipole-dipole interactions. Hydrogen bonding is responsible for many remarkable properties and behaviors of materials, including high boiling and melting points of water, its surface tension, and its ability to dissolve an array of polar and ionic compounds.
Hydrogen bonding plays a pivotal role in biological molecules like DNA, proteins, and RNA; its stability and interactions play an essential part of their life cycles and interactions. Furthermore, this phenomenon provides important insight into molecular behavior as well as the properties of various substances.
Key Difference Between Dipole-Dipole Interactions and Hydrogen Bonding
The main difference between dipole-dipole interactions as well as hydrogen bonding lies in the fact that dipole interactions take place when partially negatively charged molecules come into contact with another molecule that has positively charged molecules, while hydrogen bonding is a distinct form of strong dipole-dipole bonds that take place between N, F and H atoms.
Here’s a comparison chart highlighting how molecular structure affects Dipole-Dipole Interactions and Hydrogen Bonding:
| Aspect | Dipole-Dipole Interactions | Hydrogen Bonding |
|---|---|---|
| Nature of Interaction | Occurs between polar molecules. | A specific type of dipole-dipole interaction involves a hydrogen atom bonded to a highly electronegative atom (N, O, F). |
| Influence of Molecular Polarity | Affected by the degree of polarity in molecules. Larger electronegativity differences result in stronger dipole moments. | Requires a highly electronegative atom (H-bond donor) covalently bonded to hydrogen and an electronegative atom (H-bond acceptor) in another molecule. |
| Molecular Shape | The spatial arrangement of atoms can affect the overall polarity of the molecule and, consequently, the strength of dipole-dipole interactions. | The arrangement of the hydrogen atom, hydrogen bond donor, and hydrogen bond acceptor atoms is crucial for the formation and strength of hydrogen bonds. |
| Role of Dipole Moments | Larger dipole moments (resulting from greater charge separation) lead to stronger dipole-dipole interactions. | Hydrogen bonds rely on the dipole moments created by the electronegative atom (H-bond donor) and its interaction with an electronegative atom (H-bond acceptor). |
| Examples | Molecules like hydrogen chloride (HCl) and chloroform (CHCl3) exhibit dipole-dipole interactions. | Substances like water (H2O), ammonia (NH |
Comparing the Strengths of Dipole-Dipole Interactions and Hydrogen Bonding
Interaction of Dipole-Dipole Systems:
- Strength: Dipole-dipole interactions tend to be stronger than most intermolecular forces but are weaker than covalent bonds.
- Nature: Polar molecules attract one another due to partial positive and negative charges (dipoles) within these molecules.
- Magnitude of Strength: The magnitude of dipole-dipole interactions depends on the magnitude of molecular dipole moments, which in turn are determined by factors such as differences in electronegativity between atoms and molecular geometry.
- Example: Substances such as hydrogen chloride (HCl) and chloroform (CHCl3) exhibit dipole-dipole interactions.
- Dipole-dipole interaction: It plays a significant role in polar compounds’ properties such as higher boiling and melting points, their solubility in polar solvents, and other physical characteristics.
Hydrogen Bonding:
- Strength: Hydrogen bonding is one of the strongest intermolecular forces.
- Nature: Dipole-dipole interactions occur when hydrogen bonds with highly electronegative atoms such as N, O or F in one molecule to form bonds with equally electronegative atoms in another molecule forming dipole bonds between these atoms.
- Magnitude of Strength: The magnitude of hydrogen bonding strength depends primarily on its electronegativity and how effectively hydrogen acts as a bridge between all involved atoms.
- Example: Substances such as water (H2O), ammonia (NH3) and hydrogen fluoride (HF) all exhibit hydrogen bonding due to their combination of highly electronegative atoms with hydrogen atoms bonded together via hydrogen bonds.
Hydrogen bonding plays an integral part in many remarkable properties found in substances, such as its high boiling and melting points, surface tension and ability to dissolve various polar and ionic compounds. Furthermore, hydrogen bonding plays an essential role in biological processes like DNA base pairing and protein folding.
The Role of Dipole-Dipole Interactions and Hydrogen Bonding in Biological Systems
Hydrogen bonding and dipole-dipole interactions are key forces that are crucial to biological systems. They affect the structure, stability, and interplay of biomolecules.
Dipole-dipole interaction, which results from the positive and negative charges in polar molecules plays a role in the folding of proteins the enzyme-substrate interplay, and interactions between molecules such as DNA and DNA. These interactions are essential to the 3-D structure of proteins and ensure their correct function.
They also are essential to the structure of bilayers of lipids inside cell membranes. This creates an exclusive barrier to control the movement of molecules inside and out of cells.
Hydrogen bonding is one of the strongest forms of dipole-dipole interactions is essential to biological systems. It regulates the base pairing that is complementary to DNA that is where hydrogen bonds are formed between Adenine (A) and Thymine (T) along with as the guanine (G) and Cytosine (C) to help maintain the double-helix form.
Hydrogen bonding in proteins helps stabilize secondary structures such as beta sheets and alpha-helices and beta sheets, which affect the overall structure. In addition, hydrogen bonds play a vital in forming bonds between water molecules to biomolecules helping to solve the problem and aiding in various biochemical reactions.
Dipole-dipole interaction and hydrogen bonding are vital biological forces that shape the form and nature of biomolecules. They allow the preciseness and flexibility required by life processes that range from the storage of genetic information within DNA to enzymes’ catalytic activities and their crucial role in the complex mechanism of living organisms.
How Molecular Structure Affects Dipole Dipole Interactions and Hydrogen Bonding
Dipole-Dipole Interactions:
- Polarity of Molecules: Dipole-dipole interactions occur among polar molecules. Their level of polarity depends on electronegativity differences among its constituent atoms those with more significant differences have stronger dipole-dipole interactions and display stronger dipole-dipole effects.
- Molecular Structure: The spatial arrangement of atoms within a molecule determines both its charge distribution and strength of dipole moments, impacting how strongly these dipole moments act as forces between adjacent dipole moments. Even if a molecule contains polar bonds, its overall geometry could result in partial cancellation of these dipole moments, decreasing the strength of dipole-dipole interactions between dipole moments; for instance, linear molecules might exhibit stronger dipole-dipole interactions than bent or symmetrical molecules.
- Size of Dipole Moments: The magnitude of dipole moments depends on bond polarity and distance between charges; longer bonds with larger differences in electronegativity produce larger dipole moments, leading to stronger dipole-dipole interactions.
- Hydrogen Bonding: Atoms Required for Hydrogen Bond Donor and Acceptor Spaces: For hydrogen bonding to occur, molecules must contain at least one hydrogen atom bonded with an electronegative atom such as N, O or F that serves as the donor of hydrogen bonds, while another molecule contains another such electronegative atom that acts as acceptor, their exact placement within a molecule is essential to its successful functioning.
- Distance and Angle: The relationship between hydrogen, its donor atom, and its acceptor atom is extremely significant for the strength of hydrogen bonding. When they come closer and align more closely in orientation, stronger hydrogen bonds form; an example would be water (H2O), where its linear arrangement between two oxygen atoms and hydrogen results in strong hydrogen bonding.
- Number of Hydrogen Bonds: Some molecules can form multiple hydrogen bonds simultaneously, which strengthen intermolecular forces overall. For instance, water molecules possess two hydrogen atoms which may form hydrogen bonds with adjacent water molecules, creating a network of intermolecular forces and strengthening intermolecular forces further.
Solubility and Intermolecular Forces of Dipole Dipole Interactions and Hydrogen Bonding
Solubility, an integral concept in chemistry, refers to the ability of one substance (the solute) to dissolve in another substance (the solvent) to form a homogeneous solution.
Solubility research is of great significance as it underpins various chemical processes, industrial applications, and environmental concerns. Intermolecular forces play an integral part in solubility studies dipole-dipole interactions and hydrogen bonding being two significant contributors.
Dipole-dipole interactions play a pivotal role in determining solubility when working with polar solvents and solutes, particularly when they contain partial positive and negative charges that create attractive forces to facilitate dissolution processes. When water molecules come in contact with polar ions or molecules, dipole-dipole interactions foster their dispersion and dissolution processes.
On the other hand, hydrogen bonding, an intense form of dipole-dipole interaction, can have an enormous effect on solubility. Water, an immensely common solvent on earth, owes its remarkable solubilizing powers to an extensive network of hydrogen bonds formed with polar and ionic substances through hydrogen bonding.
This network enhances solubility for compounds like salts, alcohols, and organic acids dissolved into its solution. Hydrophobic interactions play an integral part in dissolving biomolecules like DNA and proteins into their solution environment.
Comparing hydrogen bonding to dipole-dipole interactions reveals that hydrogen bonding is stronger, making it more effective at dissolving various substances. Furthermore, molecular structure, temperature, and pressure all play different roles when considering solubility; hydrogen bonding exhibits unique behavior depending on these conditions.
“How are Dipole Dipole Interactions and Hydrogen Bonding Disrupted?”
Dipole-dipole interactions and hydrogen bonding can be broken by various external influences and conditions, including changes that reduce or remove attractive forces between molecules.
Some methods by which dipole-dipole interactions and hydrogen bonds may be affected are:
- Temperature: An increase in temperature gives molecules additional kinetic energy that can overcome intermolecular forces and erode dipole-dipole interactions, weaken hydrogen bonds, and ultimately bring about the phase transition from liquid to gas state.
- Pressure: High pressures may alter the molecular arrangements and weaken intermolecular forces in gases and liquids, including gases with strong hydrogen bonds; even at very high pressures they become less stable than before.
- Solvent Choice: Shifting between different solvents can disrupt dipole-dipole interactions and hydrogen bonding, for instance switching from one that facilitates them (polar) to one that does not facilitate these bonds (nonpolar) can weaken or break them altogether.
- Diluting: Diluting a solution by adding more solvent dilutes its solute molecules and can weaken interactions among solute particles, ultimately leading to their disruption.
- Chemical Reactions: Certain chemical reactions have the ability to disrupt hydrogen bonds and dipole-dipole interactions in organic molecules. Hydrolysis reactions involve water molecules dissolving hydrogen bonds within organic molecules.
- pH Changes: Varying the pH in any solution can interfere with hydrogen bonding in biomolecules like proteins and nucleic acids, potentially altering their ability to bond efficiently together. Extreme changes to pH may even denature proteins by disrupting hydrogen bonds within secondary or tertiary structures of their secondary and tertiary structures.
- Increased Ionic Strength: Elevated concentrations of ions can disrupt hydrogen bonds and dipole-dipole interactions as these interactions become stronger with water molecules than between themselves.
Summary
Dipole-Dipole Interactions and Hydrogen Bonding are intermolecular forces that exert an impactful effect on molecules’ behavior and properties. Dipole-dipole interactions form when two polar molecules attract each other through the attraction between their partial positive and negative charges.
Hydrogen bonding is an especially strong dipole-dipole interaction that involves one hydrogen atom from one molecule being bound to an extremely electronegative element (N, O, or F) and forming bonds with an equally electronegative element in another molecule.
Hydrogen bonding is significantly stronger than typical dipole-dipole interactions and plays a crucial role in biological and chemical processes, including boiling, melting points, solubility, molecular stability, etc. Understanding these intermolecular forces is essential for understanding various phenomena across chemistry, biology, materials science, etc.