Introduction to BCR-ABL p190 and p210
BCR-ABL p190 and p210 are variants of an ABL gene created through translocations that is commonly linked with leukemia; specifically Chronic Myeloid Leukemia in its case for p190 while in Philadelphia Chromosome Positive Acute Lymphoblastic Leukemia for p210. Breakpoints within BCR and ABL genes vary, altering protein structure and downstream signaling pathways to create distinct symptoms in patients that affect prognosis, treatment strategies, and strategies for detection by specific molecular methods.
Targeted therapies have shown promise for improving results; however, resistance issues persist. Recognizing these variants is crucial to accurate diagnosis and effective therapy as ongoing research seeks new strategies that could enhance patient health outcomes.
Overview of BCR-ABL p190
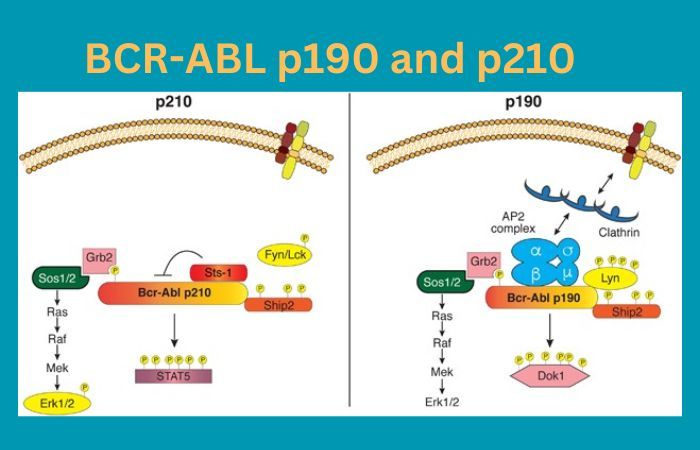
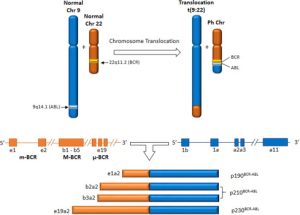
BCR-ABLp190 is one variant of the BCR-ABL fusion gene, an abnormal genetic aberration implicated in various forms of leukemia, often Chronic Myeloid Leukemia (CML). CML has its roots in Chronic Myeloid Leukemia’s primary malignancy of bone marrow cells called Chronic Myeloid Leukemia; hence its name as it produces approximately 190kDa fusion proteins from this translocation between BCR (Breakpoint Cluster Region) gene on Ch 22 and ABL1 (Abelson Tyrosine Protein Kinase 1) gene on Ch 9.
BCR-ABL p190 can have far-reaching implications on disease manifestation and progression. Clinically, it tends to manifest with more aggressive features compared to its counterpart, BCR-ABL p210; often linked with lymphoid blast crisis in CML patients and Ph+ ALL; the altered structure due to unique breakpoint location causes dysregulated signaling pathways which result in uncontrollable cell proliferation and reduced apoptosis which contribute to leukemia development.
Accurate diagnosis and monitoring of BCR-ABL p190 leukemia requires molecular techniques capable of detecting the presence of fusion transcripts bearing this marker. Tyrosine kinase inhibitors have dramatically changed treatment options available to those suffering from BCR-ABL p190-related leukemia, yet issues like resistance and recurrence remain significant concerns.
Continued research of BCR-ABL p190’s molecular mechanisms, combined with advances in personalized medicine approaches, may offer hope of refining treatment strategies and ultimately improving prognoses and quality of life for individuals living with this variant of leukemia.
Overview of BCR-ABL p210
BCR-ABL p210 is a crucial variant of the BCR-ABL fusion genes that is known for its crucial function in the development of different types of leukemia. This particular variant is specifically associated with Philadelphia Chromosome-Positive Chronic Myeloid Leukemia (CML) and certain forms are Acute Lymphoblastic Leukemia (ALL).
The term “p210” stems from the molecular mass of the fusion protein that it produces around 210 kDa result of a chromosomal transfer among the BCR (Breakpoint Cluster Region) gene on chromosome 22, along with the ABL1 (Abelson Tyrosine-Protein Kinase 1) gene on chromosome 9.
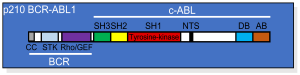
BCR-ABL p210 has profound effects on cell signaling pathways because of its specific breakpoint location leading to uncontrolled cell proliferation, decreased Apoptosis, and the genesis of leukemia.
The clinical manifestation of this variant is different from other variants of BCR-ABL, with a chronic phase that may develop into blast and accelerated stages, which could lead to serious complications if untreated. The diagnosis and tracking of BCR-ABL Leukemias that are p210-related require molecular methods to determine P210 transcription factors that are fusion.
The advancements in targeted therapies, specifically tyrosine kinase inhibitors have changed the treatment paradigm for patients suffering from BCR-ABL p210-positive leukemia. These treatments have dramatically improved the outcomes of patients by specifically reducing the fusion protein’s maladaptive activity. Despite these advances, obstacles like drug resistance and the progression of disease remain major obstacles.
The Clinical Features of BCR-ABL p190 and p210
Clinical features associated with BCR-ABL variants including the p190 and p210 versions:
Clinical Features of BCR-ABL P190:
- Leukemia Type: BCR-ABL p190 has been found most commonly associated with Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia (Ph+ ALL) as well as certain Chronic Myeloid Leukemia (CML) cases within their lymphoid blast crisis phase.
- Age of Onset: Ph+ ALL with p190 fusion tends to occur more commonly among adults; it has also been documented among children and adolescents. CML with P190-associated Blast Crisis typically presents at an identical age distribution as traditional CML cases.
- Symptoms: Patients diagnosed with p190-positive leukemia can exhibit many of the same signs and symptoms associated with ALL and CML, including fatigue, weakness, easy bruising and bleeding. Lymphadenopathy and splenomegaly may also develop.
- Blast Count in CML: Fusion between p190 gene is associated with increased lymphoid blast cells during blast crisis phase of CML.
- Prognosis: Ph+ ALL cases that contain p190 fusion may also carry less favorable prognoses compared to their allele counterparts, due to p190 fusion being associated with more aggressive behaviors and poorer prognoses during blast crisis phase.
Clinical Features of BCR-ABL P210:
- Leukemia Type: BCR-ABL p210 is most frequently linked with Chronic Myeloid Leukemia (CML), particularly during its chronic phase. However, it may also occur in some Ph+ ALL cases or CML that progresses into an accelerated or blast crisis phase.
- Age of Onset: CML with P210 Fuse typically affects adults; however it has been known to occur in both children and the elderly.
- Symptoms: Early on in their disease course, those affected with CML who test positive for P210 may only present with subtle or vague symptoms including fatigue, weight loss and night sweats. As it progresses further on its course, more obvious manifestations such as anemia, splenomegaly and increased susceptibility to infections become evident.
- Blast Crisis in CML: When transitioning into an accelerated or blast crisis phase of CML, myeloid blast cells typically increase dramatically and accelerate disease progression.
- Prognosis: Chronic phase CML with p210 fusion generally follows an indolent course compared to blast crisis phase, especially thanks to targeted therapies like tyrosine kinase inhibitors; prognosis and survival rates have improved greatly as a result of such therapies.
Treatment Approaches
The approaches to treating BCR-ABL-associated leukemias, p190 and P210 focus on targeted therapies that are significantly changing the way we manage and treatment of these illnesses. The therapies are designed to block the abnormality of protein fusions produced by the BCR-ABL gene variations, blocking the signaling pathways which trigger uncontrolled growth of cells and expansion.
Targeted Therapies:
- Tyrosine Kinase inhibitors (TKIs): TKIs are the primary treatment of BCR-ABL and related leukemias p210. Imatinib was the initial TKI that was developed and has demonstrated impressive results in bringing about an increase in survival and improving remission rates.
It targets specifically the abnormal tyrosine-kinase activity of BCR-ABL’s fusion protein and results in inhibition of signaling pathways downstream. Third-generation and second-generation TKIs like dasatinib and nilotinib, have been specifically designed to combat the resistance or intolerance of imatinib.
- Resistance Management: Despite the initial successes of TKIs however, the appearance of resistance-related mutations could reduce their effectiveness. Monitoring of the transcript levels of fusion and analyses of mutations within the kinase domain is essential in identifying resistance. Modifications to treatment, like changing to stronger TKIs or combining therapies could be used to combat resistance.
Considerations and Future Directions:
- Minimal Residual Disease (MRD) Monitoring: MRD refers to the presence of remaining leukemia cells in levels that cannot be detected using traditional techniques. Being able to achieve MRD negative results in a greater reaction to treatment, and can lead to better longer-term results. MRD screening is important in both the p190 and P210 leukemias.
- Stem cell transplantation: When TKIs do not work or fail to deliver allogeneic stem cell transplantation (bone marrow transplant) may be a viable option, especially for patients suffering from advanced stages or chronic disease.
- Personalized Medical: With our knowledge of the molecular and genetic foundation of these leukemias the possibility of personalized medical approaches is being investigated. Specific genetic mutations that go in addition to BCR-ABL the combination of treatments, may improve the outcomes of treatment.
- Clinical Trials: Continuous research as well as clinical trials are examining innovative therapies like newer versions of TKIs and combination therapies as well as immunotherapies. The trials help refine treatments and enhance patient outcomes.
Difference Between BCR-ABL p190 and p210
Here’s a simplified comparison chart outlining the key differences between BCR-ABL p190 and p210:
| Aspect | BCR-ABL p190 | BCR-ABL p210 |
|---|---|---|
| Associated Leukemia | Chronic Myeloid Leukemia (CML), Lymphoid Blast Crisis, Ph+ Acute Lymphoblastic Leukemia (ALL) | Chronic Myeloid Leukemia (CML), Some CML Cases, Acute Lymphoblastic Leukemia (ALL) |
| Fusion Protein Weight | ~190 kDa | ~210 kDa |
| Breakpoint Location | BCR gene on chromosome 22, ABL1 gene on chromosome 9 | BCR gene on chromosome 22, ABL1 gene on chromosome 9 |
| Clinical Presentation | More aggressive phenotype, lymphoid blast crisis, Ph+ ALL | Chronic phase accelerated and blast crisis phases, some CML cases |
| Molecular Consequences | Dysregulated signaling, uncontrolled cell proliferation, reduced apoptosis | Dysregulated signaling, uncontrolled cell proliferation, reduced apoptosis |
| Diagnosis & Monitoring | Detection through molecular methods for p190 fusion transcripts | Detection through molecular methods for p210 fusion transcripts |
| Treatment Approach | Targeted therapies (tyrosine kinase inhibitors) improved outcomes | Tyrosine kinase inhibitors revolutionized the treatment |
| Challenges | Drug resistance, lymphoid blast crisis, aggressive course | Drug resistance, disease progression, potential acute phases |
| Prognosis | Generally poorer prognosis, aggressive course | The chronic phase often precedes accelerated or blast crisis phases |
| Research Focus | Variant-specific mechanisms, potential treatments | Molecular mechanisms underlying p210-related leukemias |
| Aspect | Details about BCR-ABL p190 | Details about BCR-ABL p210 |
Similarities – BCR-ABL p190 and p210
BCR-ABL p190 and p210 genes exhibit some strikingly similar properties due to their common origin as fusion genes resulting from chromosomal translocations. Both variants play an essential role in leukemia development, contributing to both Chronic Myeloid Leukemia (CML) and Acute Lymphoblastic Leukemia (ALL).
They result from translocations between two genes on different chromosomes (BCR (Breakpoint Cluster Region) on Ch 22 and ABL1 (Abelson Tyrosine Protein Kinase 1), leading to the combination of their genetic material. Fusion produces hybrid proteins with altered structures, leading to dysregulated cell signaling pathways and ultimately contributing to uncontrollable cell proliferation, reduced apoptosis, and the progression of leukemia in humans.
Diagnostic and monitoring techniques of both variants depend on molecular methods that identify fusion transcripts to ensure accurate disease identification and monitoring.
Targeted therapies, especially tyrosine kinase inhibitors, have revolutionized treatment outcomes among those suffering from either p190 or p210 leukemia by targeting their abnormal fusion proteins’ activity; yet both variants still pose challenges such as drug resistance; thus necessitating continued research into novel approaches for therapy.
At its core, both BCR-ABL p190 and p210 variants of the cancer possess similar molecular foundations that interfere with normal cell mechanisms, leading to disease presentation and progression. Targeted therapies have greatly advanced treatment plans for either variant.
Ending
BCR-ABL p190 and p210 are two variants of the BCR-ABL fusion gene associated with specific forms of leukemia, specifically Ph+ ALL and CML. P190 tends to cause aggressive forms while P210 typically appears with CML; both varieties cause uncontrollable cell growth with decreased apoptosis rates.
Treatment involves targeted therapies (tyrosine kinase inhibitors are most often recommended), molecular monitoring is critical in tracking effectiveness as well as resistance, and ongoing research can offer hope in refining treatment strategies while improving patient outcomes.