The primary distinction between polarizability and dipole moments is the fact that polarizability can be described as an indicator of the degree to which an electron cloud can be altered due to the electrical field while dipole moment is the process of separating of negative and positive charge within a system.
The dipole moment and the polarizability are two key terms in chemistry. They are explained with respect to atoms, molecules, and electron clouds.
What is Polarizability?
The term “polarizability” can be defined as the capacity of matter to develop an electric dipole in relation to the field that is applied to the field of electricity. It is an inherent property of every matter when we think that matter is made up of particles that possess the electric charge through the use of electrons and protons.
With the help of an electric field electrons that are negatively charged and positively charged nuclei of atomic matter suffer the influence of opposite forces and charges. Polarizability plays a role in an increase in the dielectric constant of the substance and the refractive index if high frequencies are taken into consideration.
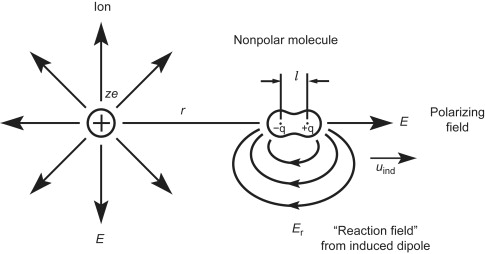
The polarizability for molecules or an atom is determined by the ratio between the dipole moment induced by the field of electric current in the local area. If a crystallized solid is considered, it’s possible to detect the dipole moments per unit cell.
The local electric field may be measured by a molecule, which generally is different from the microelectronic field we are able to observe from the outside.
In the same way, magnetic polarizability is related to the potential of the dipole moment of magnetic energy to be in relation to the external magnetic field. Magnetic polarizability and electric polarizability influence the reaction of a bound system to external magnetic fields. They also provide an insight into the structure of the molecules.
What is a Dipole Moment?
The dipole moment is an essential concept in chemistry that defines the polarity of molecules. It represents the separation of positive and negative charges within molecules to form an electric dipole; this separation results in a quantifiable vector quantity called the dipole moment; usually symbolized as μ.
Dipole moments are calculated by multiplying the magnitude of charge separation (difference in electronegativity between atoms) by the distance between charges; mathematically speaking, dipole moments equal μ = q × r, where q is charge separation and r is distance.
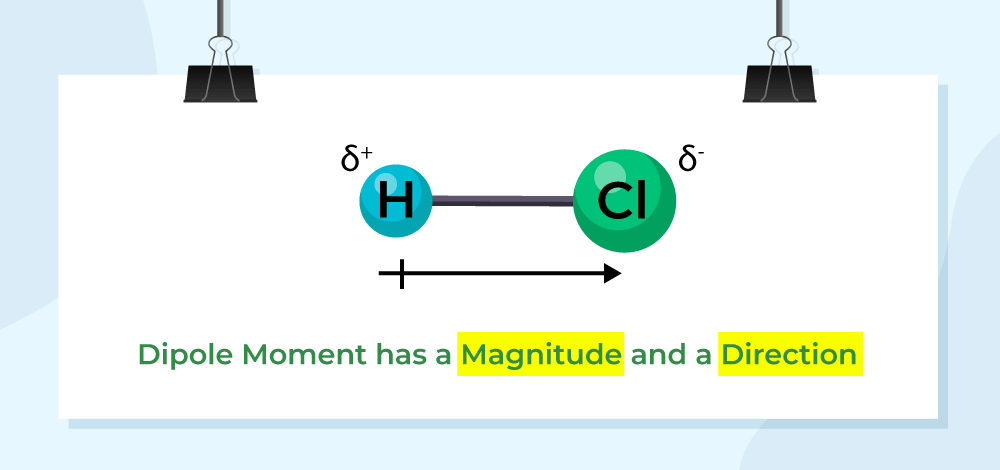
Polar molecules have an uneven distribution of electrons that leads to positive and negative ends with the positive being where electrons are pulled away and concentrated at their respective places respectively. Conversely, molecules with symmetrical electron distribution and zero dipole moment are considered nonpolar.
Dipole moments play an essential part in understanding various chemical phenomena, such as molecular polarity, intermolecular forces, and chemical reactivity. Dipole moments affect a molecule’s behavior in solvents, its boiling and melting points as well as interactions with other charged or polar molecules.
Dipole moments find applications in fields such as spectroscopy and materials science where an understanding of molecular polarity is integral for interpreting experimental data or designing materials with specific properties.
The Importance of Polarizability and Dipole Moment in Predicting Molecular Behavior
Importance of Polarizability:
- Intermolecular Forces: Polarizability is an integral element in London dispersion forces (van der Waals forces). Molecules with higher polarizability experience stronger dispersion forces that attract their constituent molecules more strongly, leading to greater attraction between molecules.
- Solubility: Polarizability affects a molecule’s solubility in nonpolar solvents. Molecules with higher polarizability tend to form temporary dipoles that interact favorably with nonpolar molecules and increase solubility.
- Chemical Reactivity: Polarizability can produce temporary dipoles that trigger different chemical interactions in chemical reactions, including London dispersion forces, nucleophilic attacks, and dipole-induced dipole interactions. Predicting how polarizability influences these reactions is essential to understanding reaction mechanisms.
- Materials Science: Polarizability is essential in materials science for understanding the dielectric properties of materials, particularly their response to electric fields, as it plays an integral part in designing capacitors, insulators, and other electronic components.
- Spectroscopy: Polarizability plays an integral part in interpreting spectroscopic techniques like Raman spectroscopy and polarimetry, which use electromagnetic radiation to interact with molecules with polarizable bonds – helping identify substances by their spectrums.
Importance of Dipole Moment:
- Dipole Moment as an Indicator of Polarity: Knowing whether a molecule is polar or nonpolar by its dipole moment is critical in understanding its behavior in chemical reactions, solubility studies, and intermolecular interactions.
- Intermolecular Interactions: Dipole-dipole interactions among polar molecules play a critical role in shaping properties such as boiling points, melting points, vapor pressure, and solubility of substances under various conditions. Predicting their strength helps shed light on their behavior in various settings.
- Chemical Bonding: Dipole moments provide insight into the nature of chemical bonds. They help assess charge separation in covalent bonds as well as overall polarity of molecules.
- Biochemical Research: Dipole moments are central to understanding the structures and functions of biomolecules like proteins and DNA. Predicting dipole moments can shed light on molecular folding, receptor interactions, and biological processes.
- Materials Design: When it comes to designing materials, dipole moments can be modified in order to produce materials with specific properties, essential for applications like electronics, sensors, and advanced materials. This field utilizes dipole moments in such ways for material development purposes.
Investigating the Factors that Influence Polarizability and Dipole Moment
Factors Impacting Polarizability:
- Size of Molecule: Larger molecules tend to have greater polarizability due to having more electrons in their electron clouds, making them easier to distort.
- Electron Cloud Distribution: Electron distribution within molecules plays a vital role. Electrons that are loosely held or further from their nuclei tend to be more easily polarizable and must therefore be located closer together for efficient functioning.
- Number of Electrons: Atoms or molecules with more electrons tend to exhibit greater polarizability because more electrons can be bent out of shape for distortion purposes.
- Atomic or Molecular Identity: Polarizability can vary between elements and compounds; noble gases exhibit lower polarizability due to their small sizes and stable electron configurations.
Dipole Moment Affecting Factors:
- Electronegativity Difference: Electronegativity differences among atoms within a molecule play an enormous role in its dipole moment; larger electronegativity disparities lead to stronger dipole moments.
- Molecular Geometry: Atoms’ arrangement within molecules determines their dipole moment. Molecules with an asymmetric geometry tend to exhibit higher dipole moments than their symmetrical counterparts.
- Charge Separation: Dipole moment is directly related to the magnitude of charge separation within molecules; greater charge separation leads to greater dipole moment.
- Bond Polarity: Polar covalent bonds contribute to a molecule’s overall dipole moment while nonpolar molecules feature electron sharing that is equal or nearly equal between bonds.
- Presence of Lone Pairs: Molecules with free electron pairs on their central atom can exhibit dipole moments that vary greatly, depending on their geometry. This may alter their dipole moment effect significantly.
- Hybridization: The type of hybridization found within a molecule’s central atom can have a substantial impact on its dipole moment. Different hybridization states result in various degrees of charge distribution.
Key Difference Between Polarizability and Dipole Moment
Polarizability and dipole moments are two terms that have significance. The main difference between dipole moment and polarizability is that polarizability is a measurement of the speed at which an electron cloud is altered through an electrical field while dipole moment is the distinction between negative and positive charges in an entire system.
Here’s a comparison chart summarizing the key differences between polarizability and dipole moment:
| Aspect | Polarizability | Dipole Moment |
|---|---|---|
| Definition | The ability of an atom or molecule to be distorted by an external electric field. | A measure of the separation of positive and negative charges within a molecule. |
| Nature of Property | A scalar property, typically expressed in cubic angstroms (ų). | A vector property, typically expressed in Debye units (D). |
| Factors Affecting | Influenced by the size and electron cloud distribution of the atom or molecule. | Influenced by the difference in electronegativity and bond geometry within a molecule. |
| Significance | Relates to the ease of inducing temporary dipoles (London dispersion forces) and affects intermolecular forces. | Determines the polarity of a molecule and its interactions with other polar molecules. |
| Measurement Techniques | Determined through experimental methods like dielectric constant measurements. | Calculated based on charge separation and bond distances within a molecule. |
| Examples | Noble gases have low polarizability due to their small size. | Water has a significant dipole moment due to its bent molecular geometry and differences in electronegativity. |
| Units and Symbols | Typically expressed in ų, sometimes denoted as “α.” | Typically expressed in D (Debye units), represented by the symbol “μ.” |
| Relationship to Chemistry | Important to understand van der Waals forces, dispersion forces, and the behavior of nonpolar substances. | Essential for understanding molecular polarity, solubility, and intermolecular interactions in polar substances. |
Similarities Between Polarizability and Dipole Moment
Polarizability and Dipole Moment: Two properties associated with individual molecules provide insight into their characteristics and behavior in various chemical contexts.
Measured Units: Both properties can be measured using units of measurement. Polarizability can typically be expressed in cubic angstroms (A3) while dipole moment is typically assessed in Debye units (D).
Relevance to Intermolecular Forces: Both polarizability and dipole moment have significant implications for intermolecular forces, with polarizability having an impactful London dispersion force (van der Waals forces), which play an essential role in all molecules; dipole moment plays a decisive role in determining dipole-dipole interactions that have strength.
Impact on Physical Properties: Both properties can have an effect on the physical properties of substances. Polarizability may alter properties like melting and boiling points for nonpolar substances; dipole moment contributes to properties like polarity, solubility and boiling points for polar substances.
Experimental Measurement: Polarizability and dipole moments can both be experimentally measured using various techniques, enabling scientists to accurately understand these properties for specific molecules.
Applications of Polarizability and Dipole Moment in Chemistry and Physics
Applications of Polarizability:
- Intermolecular Forces: Polarizability plays an integral part in understanding London dispersion forces (van der Waals forces). It determines the strength of attraction between molecules and affects properties like boiling points and phase changes.
- Solubility: Polarizability has an influence on the solubility of molecules in nonpolar solvents; nonpolar molecules with higher polarizability tend to dissolve more readily in nonpolar solvents.
- Chemical Reactivity: Polarizability affects chemical reactions involving species with temporary dipoles, such as nucleophilic attacks and certain forms of chemical bonding.
- Material Properties: Polarizability in materials science is key to understanding dielectric properties, which influence electric fields as well as applications like capacitors and insulators.
- Polarizability is used: In interpreting results from techniques like Raman spectroscopy and polarimetry that rely on interactions between electromagnetic radiation and polarizable molecules.
Applications of Dipole Moment:
- Dipole Moment as an Indicator of Molar Polarity: Dipole moment is an excellent measure of molecular polarity. It helps determine whether or not a molecule is polar or nonpolar, with implications for its chemical behavior and interactions.
- Intermolecular Interactions: Dipole-dipole interactions among polar molecules play a pivotal role in explaining various phenomena, including liquid and solution properties such as boiling points, vapor pressures and solubilities. They exert an impactful force that ultimately determines these properties.
- Chemical Bonding: Dipole moments provide invaluable insight into the nature of chemical bonds. In molecules with covalent bonds containing polar molecules, dipole moments help characterize charge separation levels.
- Dipole Moments in Biochemistry: Dipole moments play a pivotal role in understanding the structure and function of biomolecules like proteins and DNA. Dipole moments help ensure their folding and stability.
- Materials Design: Dipole moments in materials science can be harnessed to design materials with specific properties, such as ferroelectricity used in electronic devices.
- Spectroscopy: Dipole moments can be used as an interpretive tool in various spectroscopic techniques, including infrared (IR) spectroscopy where vibrational modes can be identified based on changes in dipole moments associated with molecular vibrations.
Measuring Polarizability and Dipole Moment: Techniques and Methods
Measuring Polarizability:
- Dielectric Constant Measures: Dielectric Constant measurements provide scientists with an efficient way to quantify how well materials store electrical energy within an electric field, thus providing a means for polarizability measurements on gases and liquids. This technique is usually utilized by scientists when investigating substances like gases or liquids.
- Refractive Index Measurements: The refractive index is one method for gauging the polarizability of materials. Changes in refractive index with various conditions like temperature or pressure provide insight into polarizability; this method is especially beneficial when measuring transparent substances.
- Molecular Dynamics Simulations: Computational techniques like molecular dynamics simulations can be used to calculate polarizability by simulating how atoms and molecules respond to external electric fields.
- Raman Spectroscopy: Raman spectroscopy measures the inelastic scattering of light by molecules. Polarizability influences Raman scattering intensity, so an analysis of Raman spectra can give insight into its properties.
Measuring Dipole Moment:
- Dielectric Spectroscopy: Dielectric spectroscopy can be used to accurately assess the dipole moment of molecules. By subjecting samples to an alternating electric field and monitoring their response, researchers can quickly establish both its magnitude and orientation within their sample.
- Infrared (IR) Spectroscopy: IR spectroscopy can detect molecular vibrations such as stretching and bending modes in chemical bonds, as well as differences between ground states of molecules and their excited states that can be inferred from IR spectra.
- Nuclear Magnetic Resonance (NMR) Spectroscopy: NMR is used to study nuclear spin. Dipole-dipole interactions between nuclear spins allow researchers to measure molecular dipole moments. NMR is particularly useful for studying organic molecules’ dipole moments.
- Electron Paramagnetic Resonance (EPR) Spectroscopy: EPR spectroscopy is used to analyze the response of unpaired electrons in paramagnetic substances to an applied magnetic field. It can be used to study radical dipole moments as well as other paramagnetic species.
- Electric Field Gradients (EFGs) in NMR: Certain NMR experiments allow researchers to measure the electric field gradients experienced by nuclei within molecules during magnetic resonance imaging (MRI). These gradients are affected by molecular dipole moments and local electron densities.
- Dipole Moment Calculations: Quantum mechanical calculations such as density functional theory (DFT) or ab initio methods can be used to compute dipole moments based on molecules’ electronic structures and determine their dipole moments.
Summary
Polarizability measures the ease with which an atom or molecule can be altered by an external electric field, most often by London dispersion forces in nonpolar substances.
It’s expressed as a scalar property expressed in cubic angstroms (A3). Dipole moment, by contrast, measures the separation between positive and negative charges within a molecule, thus determining its polarity and interactions with other polar molecules.
The dipole moment is typically measured in Debye units (D). Polarizability depends on factors like size and electron distribution; dipole moment draws upon electronegativity differences and molecular geometry.
Polarizability provides crucial insight into nonpolar substance behavior while dipole moment helps reveal molecular polarity, solubility, and interactions among polar molecules.

