Hydrogen Bond and Ionic Bond
Hydrogen Bond and Ionic Bond both involve electrostatic attractions between charged particles.
Hydrogen Bond: A hydrogen bond forms between hydrogen atoms and highly electronegative elements like oxygen, fluorine or nitrogen – such as oxygen fluorine or nitrogen – via their partial charges interacting with each other to form the interaction that results in this type of relationship. Hydrogen bonds play an integral part of life processes such as water boiling point surface tension DNA stability protein folding. Though weaker than their counterpart ionic or covalent bonds they play an essential part.
Ionic Bond: The formation of an ionic bond occurs when two atoms possess significantly differing electronegativities, leading to electron transference between them and leads them into each other’s orbitals, eventually becoming positively or negatively charged ions with oppositely charged charges attracted together through strong electrostatic forces forming strong electrostatic forces attracting each pair together.
Eventually creating the bonds known as an ionic compound, typically distinguished by crystal structures with high melting points, boiling temperatures and boiling points, often used as main constituents in salts as well as important roles chemical reactions as well as stabilizing mineral structures. Ionic bonds, on the other hand, involve electrostatic attraction between oppositely charged ions that result from electron transfer and are stable with distinct properties; they contribute significantly to both water’s characteristics as well as those of biological molecules.
Importance of understanding different types of chemical bonds
It is important to understand the different chemical bonds for several reasons:
- Structure and Properties: Chemical Bonds determine how atoms are arranged in compounds and molecules, and this in turn affects their chemical and physical properties. The characteristics of covalent, metallic, and ionic bonds influence the behaviour of substances.
- Bond Formation and Energy: Understanding chemical bonding helps us understand the process by which atoms combine to create molecules and compounds. The different bonding types are characterized by the transfer or sharing of electrons. Each type has its own energy. Understanding the bonding process and its energy can help predict reaction stability, likelihood and the changes in energy involved.
- Biological systems: Chemical bonds play a fundamental role in the structure and functioning of biological systems. The three-dimensional interaction and structure of enzymes, DNA and proteins is influenced by these bonds. The types of chemical bonding involved in biological processes can help unravel mechanisms of life and drug interactions.
- Material science and engineering: Chemical bonds affect the properties of various materials in different industries. Understanding the bonds in metals can help explain their thermal and electrical conductivity. Knowledge of covalent and ionic bondings is also essential to understand ceramics and other polymers. It is important to have this knowledge in order to design new materials that possess desired properties.
- Predicting Reactivity and Bonding Patterns: It is possible to predict reactivity by understanding chemical bonds. We can also determine how substances will react with each other. This helps to explain periodic trends, like the differences between electronegativity and reactivity in alkali metals.
Understanding different types of chemical bond is crucial for understanding the properties, behavior, reactivity and structure of substances across various scientific disciplines. We can make better predictions, develop new materials and get a deeper understanding of biological systems.
Definition of Hydrogen Bond
Hydrogen bonds are chemical bonds formed between hydrogen atoms and elements displaying negative charges such as oxygen, nitrogen or fluorine atoms that exhibit electronegative charges. Although weak in attraction strength, hydrogen bonds play an essential part in numerous biological and chemical processes.
Hydrogen bonds form when hydrogen has an electronegativity level below 1 and has a partial positive charge due to low electronegativity, attracted to an electron-dense region on an electronegative atom such as oxygen, nitrogen or fluorine in different molecules or within itself that has partial negative charges that attract it – this electrostatic attraction between positive hydrogen atoms and these electronegative ones forms the hydrogen bond.
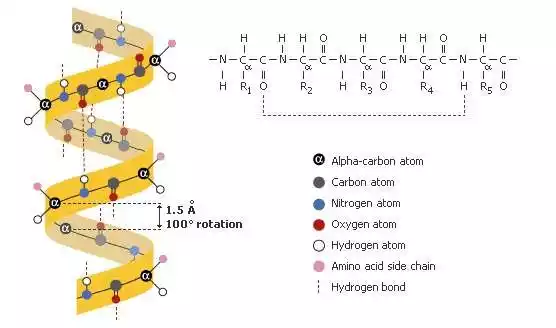
Hydrogen bonds play an essential role in nature. For instance, they contribute to water’s unique properties – its high boiling point, surface tension and ability to dissolve various substances – as well as in biological systems by maintaining DNA structure and protein composition as well as acting in molecular recognition and binding processes.
Hydrogen bonds may be weaker than covalent or ionic bonds, yet still play an integral part in determining molecular behavior and properties. Their presence or absence has the ability to greatly impact the physical and chemical characteristics of substances; understanding hydrogen bonds is therefore integral for understanding structures and behavior of molecules found across fields like chemistry, biochemistry and materials science.
Definition of Ionic Bond
An ionic bond is a form of chemical bond formed between atoms with significantly differing electronegativities that involves electron transfer from one atom to the next, creating positively charged cations and negatively charged anions.
An ionic bond occurs when one atom loses electrons to become positively charged (cation), while another gains them and forms negatively charged ions (anion). Electrostatic attraction between oppositely charged ions holds them together and forms crystal lattice structures.
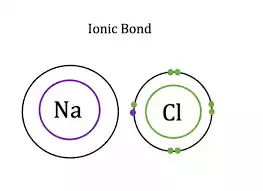
Ionic bonds can typically be seen in compounds composed of metal cations and nonmetal anions, with sodium giving up an electron to chlorine for Na+ to form Cl- ions; calcium carbonate (CaCO3) allows calcium to do this twofold and produces Ca2+ and CO32- ions as results.
Ionic compounds tend to possess higher melting and boiling points due to strong electrostatic interactions among their constituent ions, often featuring crystal structures and being excellent conductors of electricity in either their solid state or when in solution form.
Understanding ionic bonds is critical for explaining the properties and behavior of ionic compounds as well as their application across fields like materials science, chemical synthesis and electrochemistry.
Types of Hydrogen Bonds:
When discussing different kinds of hydrogen bonds, it’s essential to keep in mind the specific conditions involved as well as those belonging to each participating atom. Here are a few popular types of hydrogen bonds:
- Intermolecular Hydrogen Bonds: Intermolecular hydrogen bonds refer to bonds formed among molecules; for instance, when water (H2O) molecules come together, hydrogen atoms from one can bond to oxygen atoms on neighboring molecules by creating intermolecular hydrogen bonds between themselves and those they form intermolecular hydrogen bonds with.
- Intramolecular Hydrogen Bonds: Intramolecular hydrogen bonds form between molecules of the same molecular family; for instance, DNA forms such bonds between complementary nitrogenous bases like adenine-thymine or guanine-cytosine to keep its double helix structure together.
- Symmetric Hydrogen Bonds: Symmetric hydrogen bonds occur when two atoms share one hydrogen atom equally and contribute an electron each to form the bond, contributing an equal share to it. They tend to form in relatively uncommon systems.
- Low Barrier Hydrogen Bonds: Low-barrier hydrogen bonds have lower energy barriers for switching donor/acceptor pairs between donor and acceptor hydrogen atoms, making these important in enzyme reactions and proton transfer processes.
- Cation-Hydrogen Bond: Cation-hydrogen bonds form when hydrogen interacts with a positively charged cation; here, hydrogen acts as the donor and acceptor respectively for this form of hydrogen bonding; these connections can often be seen in molecular and crystal structures.
These are some of the various kinds of hydrogen bonds present across different molecular contexts. Each variety possesses specific attributes and effects that contribute to its unique role within chemical or biological systems.
Types of Ionic Bonds:
Ionic bonds form between atoms with significantly different electronegativities, leading to an exchange of electrons from one atom to the next. While there isn’t one “type” of ionic bonding compound or its characteristics.
Here are a few aspects related to types of ionic bonds:
- Binary Ionic Compounds: Binary ionic compounds consist of two elements — typically, one metal cation and an anionic nonmetal anion such as sodium chloride or magnesium oxide, for instance — joined together. Some examples of such ionic compounds include sodium chloride (NaCl) and magnesium oxide (MgO) being examples of binary ionic compounds.
- Polyatomic Ionic Compounds: Polyatomic ions are groups of atoms with net positive or negative charges; when combined together with other ions they form ionic compounds like ammonium nitrate (NH4NO3) in which ammonium (NH4+) bonds with nitrate ion NO3- to form its compound form.
- Transition Metal Complexes: Transition metal ions have the capacity to form ionic bonds with various other ions or ligands, creating complexes characterized by the coordination of ligands around their central metal ion for an overall ionic structure – one such complex being [Cu(NH3)4]2+ which features copper (Cu2+) forming bonds with ammonia-derived ammonia ligands (NH3) to form overall ionic structures such as [Cu(NH3)4]2+ for instance).
- Variable Charge Ions: Certain transition metal-derived ions may possess variable charges. This indicates that different metal ions exhibit various oxidation states and form bonds accordingly – for instance iron can form both Fe2+ and Fe3+ ions that form compounds like FeCl2 or FeCl3.
- Ionic Solid Solutions: Ionic compounds may form solid solutions by dissolving into different proportions within crystal lattice structures; this results in compounds with various properties or characteristics; for instance alloy formation through mixing multiple metal ions present within such solutions is one example of such outcomes.
These examples all share a similar basic principle of ionic bonding; their variations lie in terms of composition, structure and properties of the compounds formed. Noting the differences among types of ionic bonds may help us better comprehend them – they depend on specific combinations of elements that form compounds rather than distinct categories of bonding.
Comparison Table of Hydrogen Bond and Ionic Bond
Here’s a comparison chart highlighting the key differences between hydrogen bonds and ionic bonds:
| Topics | Hydrogen Bond | Ionic Bond |
|---|---|---|
| Definition | Weak electrostatic attraction between a hydrogen atom and an electronegative atom. | Strong electrostatic attraction between oppositely charged ions. |
| Formation | Occurs between a hydrogen atom and an electronegative atom in different molecules or within the same molecule. | Occurs between atoms with significantly different electronegativities. |
| Bond Strength | Relatively weak | Relatively strong |
| Bond Type | Intermolecular | Intramolecular |
| Electron Transfer | No electron transfer | Complete transfer of electrons from one atom to another |
| Nature of Attraction | Partial positive charge of the hydrogen atom attracts a partial negative charge of the electronegative atom. | Electrostatic attraction between positive and negative ions. |
| Examples | Hydrogen bonding in water molecules, DNA base pairing. | Sodium chloride (NaCl), calcium carbonate (CaCO3). |
| Physical Properties | Hydrogen bonds contribute to properties like high boiling point, surface tension, and specific molecular arrangements. | Ionic compounds tend to have high melting and boiling points and form crystalline structures. |
| Strength of Interactions | Relatively weaker than covalent or ionic bonds. | Relatively stronger than hydrogen bonds. |
| Directionality | Hydrogen bonds are directional and depend on molecular geometry. | Ionic bonds are non-directional and extend throughout the crystal lattice. |
The Similarities of hydrogen bonds and ionic bonds
Hydrogen bonds and ionic bonds differ substantially, they do share certain similarities:
- Electrostatic Attraction: Both hydrogen bonds and ionic bonds involve electrostatic interactions that draw positive and negative charges together in attraction to one another; for hydrogen bonds this occurs between partial positive charges on hydrogen atoms attracted to partial negative charges on electronegative atoms while with ionic bonds the positive charge of an anion attracts its positive counterpart, creating electrostatic force fields.
- Intermolecular Forces: Both hydrogen bonds and ionic bonds constitute intermolecular forces, occurring either between molecules or within an individual molecule and contributing to overall structure and properties of the substances involved.
- Importance in Biological Systems: Both hydrogen bonds and ionic bonds play critical roles in biological systems.
- Influence on Properties: Both types of bonds significantly alter the properties of substances they come in contact with, including hydrogen bonds contributing to specific features like high boiling point and surface tension of water, while ionic bonds play an integral part in creating crystal structures as well as producing compounds with elevated melting and boiling points.
- Hydrogen Bonding and Ionic Interaction: Both hydrogen bonds and ionic interactions play a crucial role in the solubility of substances. Hydrogen bonding between molecules may increase solubility in polar solvents while interactions between ions and solvent molecules could result in greater solubility of certain substances.
Hydrogen bonds and ionic bonds may differ in strength, formation mechanisms and nature of interactions; yet both share similar features regarding electrostatic nature, intermolecular forces, biological relevance, impact on properties and role in solubility.
Summary
In the vast realm of chemistry, hydrogen bonds and ionic bonds are pivotal forces that shape molecular structures, chemical reactions, and material properties. Their distinct yet interconnected roles underscore the complexity and elegance of the natural world. Whether in the DNA that defines life, the water that sustains it, or the compounds that power it, these bonds stand as testaments to the intricate dance of atoms and electrons.

