General and Specific Acid Base Catalysis
General and Specific Acid Base Catalysis both involve the donation or acceptance of protons to facilitate chemical reactions, but specific acid-base catalysis targets particular functional groups while general acid-base catalysis is non-specific.
Catalysis is an essential concept in chemistry that plays an integral part in speeding up chemical reactions and making them more efficient, using catalysts as vital intermediary substances to lower activation energy requirements for reactions without being consumed themselves.
Acid-base catalysis is perhaps one of the most prevalent and versatile mechanisms, using protons (H+) or hydroxide ions (OH-) donated or accepted as contributions towards reaction kinetics modulation.
Acid-base catalysis can generally be divided into two distinct subcategories, general acid-base catalysis and specific acid-base catalysis. Each category differs in terms of mechanisms, application scope, and the types of catalyst species involved knowledge of these differences helps chemists and researchers design optimal reactions in industrial processes or biological systems alike.
What is General Acid-Base Catalysis?
General acid-base catalysis refers to a catalytic mechanism in which a catalyst, typically a molecule or an ion, acts either as an acidic proton donor (an acid) or basic proton acceptor (base), to promote chemical reactions by participating in proton transfer reactions between reactants.
With this form of catalysis, its role is less specific to any particular substrate chemical structure but instead can accelerate many proton transfer reactions more broadly.
General acid-base catalysis revolves around the ability of a catalyst to alter a reaction’s energy profile by altering acidity or basicity in its environment, contributing protons either way and stabilizing transition states, lowering activation energy barriers, or speeding up rates of reaction.
This mechanism does not restrict itself to one class of chemicals but is commonly employed in both laboratory settings as well as nature for chemical reactions of all sorts.
General acid-base catalysis is an adaptable and widespread catalytic mechanism. It plays a critical role in various chemical processes, from hydrolysis reactions and enzyme reactions to chemical transformations and more, providing crucial catalytic support across an extensive variety of chemical transformations. Due to its versatility and broad applicability, general acid-base catalysis remains one of the cornerstone concepts in catalysis today.
Characteristics of General Acid-Base Catalysis?
General acid-base catalysis can be defined by several key features. These characteristics include:
General acid-base catalysis has an impressive range of applications and is suitable for various chemical processes, not merely those related to specific substrates or functional groups. Thus it makes this technique suitable in various contexts.
- Proton Transfer: Acid-base catalysis involves proton transfer reactions through its catalyst acting either as an acid donor (proton donor) or acceptor (base). This fundamental aspect is central to its catalytic mechanism.
- Intermediate Formation: Intermediates play an essential part of reaction mechanisms and catalyst-proton transfer reactions can assist by stabilizing transition states to reduce activation energy requirements for further progress of reactions.
- General Acid-Base Catalysis Doesn’t Offer As Much Precision: While general acid-base catalysis offers speed in catalyzing many reactions, specific acid-base catalysis provides greater precision.
- Potential Side Reactions: Due to its general application, general acid-base catalysis may result in unintended chemical transformations or side reactions in some instances, so careful consideration must be given in order to optimize reaction conditions and decrease undesirable outcomes.
- Examples: Common examples of general acid-base catalysis include hydrolysis of esters and base reactions catalyzed by hydroxides ions; general acids often play a part in supporting alkene-hydration reactions as part of general acid-base catalysis reactions.
- Practical Importance: Acid-base catalysis has many practical applications in various fields, from organic synthesis and industrial processes, as well as some enzymatic reactions where using more general approaches would suffice.
What is Specific Acid Base Catalysis?
Specific acid-base catalysis (or specific acid-base catalysis, for short) is a catalytic mechanism in which a catalyst, typically an active group in molecules or enzymes, facilitates chemical reactions through proton transfer reactions involving highly specific and selective reactants.
As opposed to general acid-base catalysis which covers more types of reactions simultaneously, specific acid-base catalysis specializes in catalyzing reactions involving specific functional groups or chemical moieties in substrate.
Specific acid-base catalysis stands out in its precision in recognizing and reacting with specific chemical groups within a substrate. The catalyst’s functional group has a high affinity with certain functional groups in reactants. It can donate or accept protons selectively during the catalysis of reactions that might otherwise fail without that catalytic group’s presence. Such selectivity often allows reactions that wouldn’t otherwise proceed efficiently to be catalyzed efficiently through specific acid-base catalysis.
Specific acid-base catalysis plays a crucial role in various biochemical and organic reactions. Enzyme biological catalysts often employ specific acid-base catalysis as part of their conversion of substrates into products with remarkable specificity and efficiency, using this catalytic mechanism as part of biological processes and organic synthesis research.
Keeping biological systems running is complex work requiring constant attention from researchers interested in understanding enzymatic reactions or organic syntheses as examples to keep on our radar screens.
Characteristics of Specific Acid Base Catalysis?
- High Specificity: One of the hallmarks of targeted acid-base catalysis is its remarkable specificity. A specific chemical motif or functional group in an enzyme acts as the catalytic site to interact directly with particular substrate chemical moieties for catalysis to occur effectively and selectively. Such targeted catalysis makes use of targeted recognition.
- Precise Functional Group Interaction: Certain acid-base catalysts employ precise proton transfer reactions with specific functional groups in their substrate, leading to highly controlled proton transfer reactions that often yield well-defined intermediate products.
- Specific Acid-Base Catalysis Is Highly Efficient Due to Selectivity: Specific acid-base catalysis is highly efficient due to its selectivity; by targeting certain chemical groups for interaction by its catalysts, specific acid-base catalysis significantly lowers activation energy while hastening reactions significantly faster than competing approaches.
- Limited Applicability: As opposed to general acid-base catalysis, specific acid-base catalysis is limited in its application only when reactions involve specific functional groups identified by its catalysts; hence it cannot be applied across a broad array of reactions.
- Examples: We see evidence of specific acid-base catalysis most frequently in enzyme-catalyzed reactions, where enzymes have evolved to catalyze specific reactions with great precision – examples include Chymotrypsin-mediated peptide bond cleavage and serine protease catalysis as prime examples of targeted acid-base catalysis.
- Biological Importance: Specific acid-base catalysis plays a pivotal role in biological systems, particularly enzyme-catalyzed reactions. Enzymes use acid-base catalysis to precisely control metabolic pathways for proper functioning and regulation, thus guaranteeing smooth functioning of biological processes.
- Catalyst Design and Optimization: Optimizing acid-base catalysts requires considering both substrate properties and reaction conditions when creating them, to achieve maximum effectiveness and efficiency. To get optimal performance it’s also vital that these catalysts undergo thorough tests against possible threats to their functionality – this includes toxicological analyses.
Difference Between General and Specific Acid Base Catalysis?
Here’s a comparison chart the key differences between general acid-base catalysis and specific acid-base catalysis:
| Aspect | General Acid-Base Catalysis | Specific Acid-Base Catalysis |
|---|---|---|
| Definition | Catalysis involving proton transfer reactions with broad substrate scope | Catalysis involving proton transfer reactions with specific functional group interactions |
| Catalyst Nature | Often molecules or ions act as proton donors or acceptors | Typically functional groups within molecules or enzymes |
| Substrate Range | Broad, can catalyze a wide range of reactions | Limited to reactions involving specific functional groups |
| Mechanism | Participates in proton transfer to stabilize transition states | Selectively interacts with specific functional groups |
| Specificity | Low specificity, versatile in various reactions | High specificity, targeted to particular chemical groups |
| Examples | Hydrolysis of esters, enzymatic reactions | Chymotrypsin-catalyzed peptide bond cleavage, serine protease catalysis |
| Catalytic Efficiency | Moderate efficiency due to generality | High efficiency due to specificity |
| Practical Applicability | Widely applicable in diverse chemical contexts | Useful for reactions where specific functional groups are involved |
| Limitations | Potential for side reactions, less precise | Limited to reactions with suitable functional groups |
| Catalyst Design and Optimization | Less focus on catalyst design | Requires careful design and optimization |
| Role in Nature | Less common in biological systems | Frequently employed in enzymatic reactions |
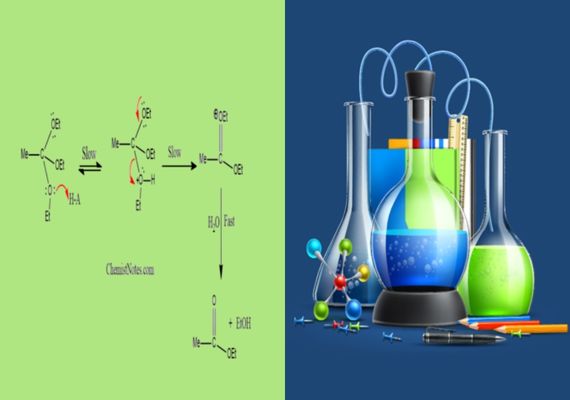
Role of Acid-Base Catalysis in Hydrolysis Reaction
Acid-base catalysis in hydrolysis reactions can be understood through three key points. These are:
- Hydrolysis Substrates Are Stable: To initiate hydrolysis reactions, substrates typically possess chemical bonds that are relatively resistant to spontaneous cleavage in water. Acid-base catalysis involves adding either an acid or base into the reaction mixture: acid can protonate certain functional groups on the substrate making them more prone to nucleophilic attack by water molecules, while the base can deprotonate specific functional groups to create nucleophilic hydroxide ions that attack electrophilic sites within it.
- Lowering Activation Energy Barrier: Acid-base catalysis lowers the activation energy barrier of hydrolysis reactions significantly faster compared to uncatalyzed reactions, as its addition stabilizes transition states of reactions, making chemical bonds break more readily and water molecules join in participating more readily in reactions.
- Specific Acid-Base Catalysis: Some hydrolysis reactions use specific acid-base catalysis, in which catalytic species target certain functional groups within a substrate to enhance efficiency and precision during reactions. Such specificity increases both efficiency and precision during these chemical transformations.
- Ester Hydrolysis: When hydrolyzing esters, both acidic and basic catalysis methods may play a part. Acid catalyzed ester hydrolysis involves protonating ester oxygen molecules to make them more vulnerable to nucleophilic attack from water molecules; while base-catalyzed ester hydrolysis utilizes deprotonating water as it deprotons to form hydroxide ions which attack ester carbonyl groups directly.
- Peptide Bond Hydrolysis: When it comes to hydrolyzing peptide bonds enzymatically, acid-base catalysis plays a critical role. Enzymes such as proteases contain catalytic residues such as serine or histidine that act both as acidic acids and bases for more targeted and efficient bond cleavage.
- Hydrolysis reactions: It plays an integral role in biological processes, from digestion and cell metabolism to DNA replication and replication itself. Enzymes with acid-base catalyzing residues play an integral part in catalyzing these hydrolysis reactions to ensure precise substrate cleavage and separation.
Role of Specific Acid-Base Catalysis in Nucleophilicity
Here’s an illustration of how specific acid-base catalysis affects nucleophilicity:
Protonation and Nucleophilicity Enhancements:
- Specific Acid Catalysis: Employing specific acids as catalysts allows them to protonate specific functional groups within their substrate and increase electrophilicity by withdrawing electron density and creating positive charges; this makes the substrate more vulnerable to nucleophilic attack at that site and enhances nucleophilicity at that site.
- Example: Hydrolyzing an ester with H3O+ acid increases its electrophilicity and facilitates nucleophilic attack from water molecules that results in ester cleavage.
Deprotonation and Production of Nucleophiles:
- Specific Base Catalysis: In specific base catalysis, the catalyst acts as a specific base by accepting proton (H+) from functional groups in a substrate, deprotonating them to produce hydroxide ions that readily participate in nucleophilic reactions and increasing electron density and nucleophilicity of deprotonated functional groups by accepting protons (H+). By extracting protons with specific base catalysis increasing electron density and nucleophilicity.
- Example: Hydrolysis of ester compounds catalyzed by certain bases such as sodium hydroxide (NaOH) involves formation of hydroxide ions with increased nucleophilicity from deprotonating water molecules to form nucleophilic hydroxides that then attack carbonyl carbons of ester molecules to cause their hydrolysis.
Control and Selectivity:
- Acid-base catalysis offers precise control and selectivity over nucleophilicity. By choosing specific acid or base catalysts with functional groups that determine which parts of a substrate become more nucleophilic or electrophilic, acid-base catalysis facilitates efficient reactions in synthetic chemistry and biochemical processes that achieve selective reactions that produce selective results.
Biological Relevance:
- Acid-base catalysis plays an integral part of many biological reactions. Enzymes use specific amino acid residues as acidic or basic catalysts to regulate nucleophilic attacks on nucleophilic attacks on proteins or nucleic acids synthesized from them by enzymes as nucleophilic attack regulators; such reactions play a vital part in DNA replication, protein synthesis and metabolic pathways among others.
Summary
General acid-base catalysis utilizes catalysts, often molecules or ions, to facilitate chemical reactions by either accepting or donating protons to chemical reactions. Although effective across multiple reactions, its lack of specificity could result in unwanted side reactions.
On the other hand, specific acid-base catalysis involves catalysts with specific functional groups within molecules or enzymes that interact specifically with substrate functional groups through proton transfer reactions.
Specific acid-base catalysis provides highly targeted catalysis that is efficient and highly targeted making it suitable for reactions involving specific chemical moieties. Specific acid-base catalysis plays a key role in biochemical processes like enzymatic reactions where precision control over reaction pathways is critical.